Amino Acids and Proteins
840 likes | 3.02k Vues
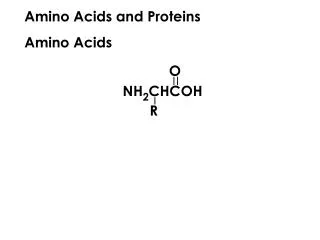
Amino Acids and Proteins. Structure of amino acids. The hydrolysis of most proteins produces about twenty different amino acids. The acids have an amino group bonded to the carbon:-. an -amino acid. Amino acid classification. The 20 amino acids are further classified:.

Amino Acids and Proteins
E N D
Presentation Transcript
Structure of amino acids The hydrolysis of most proteins produces about twenty different amino acids. The acids have an amino group bonded to the carbon:- an -amino acid
Amino acid classification The 20 amino acids are further classified: • neutral - one amino and one carboxyl group • acidic - one amino and two carboxyl groups • basic - two amino and one carboxyl group
Neutral amino acids NameSymbolStructure Alanine Ala or A CH3CH(NH2)CO2H Asparagine Asn or N H2NCOCH2CH(NH2)CO2H Cysteine Cys or C HSCH2CH(NH2)CO2H Glutamine Gln or Q H2NCOCH2CH2CH(NH2)CO2H Glycine Gly or G CH2(NH2)CO2H Isoleucine Ile or I CH3CH2CH(CH3)CH(NH2)CO2H Leucine Leu or L (CH3)2CHCH2CH(NH2)CO2H Methionine Met or M CH3SCH2CH2CH(NH2)CO2H Phenylalanine Phe or F C6H5CH2CH(NH2)CO2H Proline Pro or P
Neutral amino acids NameSymbolStructure Serine Ser or S HOCH2CH(NH2)CO2H Threonine Thr or T HOCH(CH3)CH(NH2)CO2H Tryptophane Try or W Tyrosine Tyr or Y Valine Val or V (CH3)2CHCH(NH2)CO2H
Acidic amino acid NameSymbolStructure Aspartic acid Asp or D HO2CCH2CH(NH2)CO2H Glutamic acid Glu or E HO2CCH2CH2CH(NH2)CO2H
Basic amino acids NameSymbolStructure Arginine Arg or R HN=C-H(CH2)3CH(NH2)CO2H NH2 Histidine His or H Lysine Lys or K H2N(CH2)4CH(NH2)CO2H
Essential amino acids Humans can synthesize only 10 of these amino acids. The remaining amino acids must be obtained from dietary sources and are so called essential amino acids. These are: Isoleucine, Leucine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine, Arginine, Histidine, and Lysine.
Stereochemistry of -Amino Acids Most naturally occurring amino acids have the same configuration about the carbon: L-(-)-glyceraldehyde L-(-)-serine natural amino acids
Physical Properties • high melting points with decomposition • very water soluble • insoluble in non-polar organic solvents such as ether
Isoelectric Points - the pH of a dilute aqueous solution of the amino acid at which the amino acid is exactly neutral. The isoelectric point of neutral amino acids lies between pH = 4.8 and 6.3. For the basic amino acids, it is in the range 7.8 to 10.8. The range is 2.7 to 3.2 for acidic amino acids.
Isoelectric Points At a pH above the isoelectric point, amino acids form anions; at a lower pH, protonation occurs and cations form. An amino acid is the least soluble at its isoelectric point.
Electrophoresis Electrophoresis uses isoelectric point differences to separate amino acids and proteins. • The amino acid is spotted on a strip of paper (or gel) on a center line. • The strip is moistened with a buffer solution of a given pH. • Electrodes are attached to the ends of the slip and a potential applied. • Depending on the amino acid’s charge, it will migrate either to the positive or negative electrode. The rate and direction of migration depends on the buffer pH and the isoelectric point of the amino acid.
Synthesis of amino acids - the Hell-Volhard-Zelinsky reaction alanine
Synthesis of amino acids - the Strecker synthesis NH3 adds first to give an imine then CN- attacks
The Peptide Bond The principal chain contains the amide bonds; the substituents, R, make up the side chains. The individual amino acids which make up the peptide are often referred to as amino acid residues. In some proteins, two or more polypeptide chains are joined together by disulfide bridges.
Nomenclature Starting from the free amine end of the peptide, the names of the amino acids are joined together with each amino acid being considered as a substituent of the following amino acid. The name ends with the name of the final amino acid: phenylalanylleucylthreonine Phe-Leu-Thr
Aspartame • an artificial sweetener sold under the trade name Nutrasweet O O + H N-CH-C-NH-CH-C-OCH 3 3 CH -C H CH 2 6 5 2 - CO 2 aspartylphenylalanine methyl ester Asp-Phe-OCH3 • the ester ending is indicated by -OCH3 in the abridged notation.
Angiotensin II Angiotensin II is a blood pressure regulating hormone. It contains 8 amino acid residues. It is possible to arrange these in 40,320 different ways only one of which corresponds to the hormone! Its structure is actually: Asp-Arg-Val-Tyr-Ile-His-Pro-Phe.
Sequential Degradation of Peptides Step 1: breaking disulfide linkages and purification of products Many polypeptides consist of two or more chains which are joined together by disulfide bridges. These bridges must be broken and the fragment products separated. This is done by an oxidation: chain A chain B chain A chain B
Purification The various methods used to separate polypeptides depend on size, solubility in a given solvent, charge or ability to become bonded to a support material (chromatography). • dialysis - filtration through a semi-permeable membrane • ion exchange chromatography • electrophoresis • chromatography
Step 2. What amino acids are present? The polypeptide is completely hydrolyzed (HCl 6M, 110C, 24hr) to give a mixture of the free amino acids:
Amino Acid Analysis The mixture is separated using an automated system. The apparatus includes an ion exchange column with a negatively charged support (usually carboxylate or sulfonate). The amino acid mixture, in weakly acidic solution, is added to the column. According to structure, the amino acids are more or less protonated and so will be retained more or less strongly on the column. The pH of the eluant is gradually increased which results in deprotonation and the subsequent elution of the amino acids. Elution begins with the strongest acid and ends with the weakest.
Amino Acid Analysis The eluant then passes into an analyzer which holds a special indicator, ninhydrin. The amino acid eluant forms a violet-purple color by reaction with the ninhydrin and the color’s intensity is proportional to the quantity of acid present.
Step 3. The sequential analysis of amino acids starting at the free amino end There are a number of methods used to determine the N-terminal amino acid of a peptide. They are all based on the fact that it is the only amino acid with a free amino group.
Edman Degradation Phenyl isothiocyanate, C6H5N=C=S, is a reagent which permits the progressive removal and identification of the N-terminal amino acid. The rest of the chain is not affected by the reaction therefore progressive removal is possible.
Specific Cleavage of Peptides A number of enzymes catalyze the hydrolysis of peptide bonds at specific points in an amino acid sequence. These enzymes are called proteases, peptidases or proteolytic enzymes. Trypsin is an example. It catalyzes the hydrolysis of polypeptides at the acyl group of arginine or lysine residues. Chymotrypsin cleaves at the acyl side of the aryl substituted acids phenylalanine, tyrosine and tryptophan.
C-terminal residue The enzyme carboxypeptidase cleaves the C-terminal amide bond in a peptide. The peptide is incubated with the enzyme until the first amino acid is detected.
A peptide! Treatment of a peptide with 2,4-dinitrofluorobenzene followed by hydrolysis gives N-dinitrophenylvaline. Leucine is the free acid. The peptide contains one of each of the following amino acid residues Leu, Ser, Phe, Pro, Tyr, Lys, Gly and Val. Partial hydrolysis gives four peptides which contain the indicated amino acids: peptide A:- Leu, Ser, Phe peptide B:- Ser, Pro, Tyr, Lys peptide C:- Tyr, Lys, Gly peptide D:- Lys, Val, Gly
A peptide has the following amino acid composition: 2 Arg, 2 Ile, Glu, 2 Gly, Leu, Lys, Phe, Pro, Ser, Trp Reaction with Edman’s reagent releases PTH-Leu. Trypsin, a digestive enzyme of intestinal liquids, cleaves polypeptides only at the carboxy (acid) end of arginine (Arg) and Lysine (Lys). Chymotrypsin which is also found in mammalian intestines, cleaves the carboxy end of phenylalanine (Phe), tryptophan (Trp) and tyrosine (Tyr). Hydrolysis by trypsin produces Gly-Arg, Ile-Trp-Phe-Pro-Gly-Arg, Leu-Lys, and Ser-Glu-Ile. Hydrolysis by chymotrypsin produces one peptide with a partial sequence of Leu-Lys-Gly … and another with partial sequence Phe-Pro-Gly-Arg-Ser ...