Complex Organic Molecules
This informative guide explores cellular respiration, focusing on the catabolic processes of energy production through oxidation of glucose, along with an overview of oxidative phosphorylation, glycolysis, and the citric acid cycle.


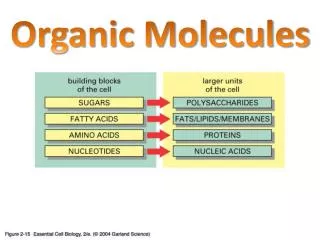
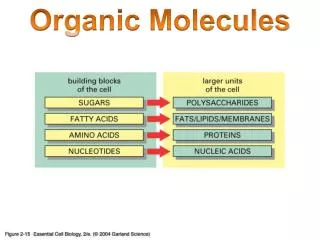
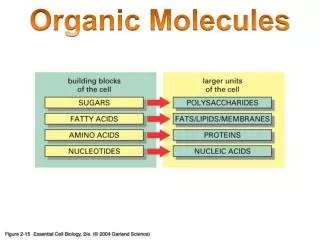
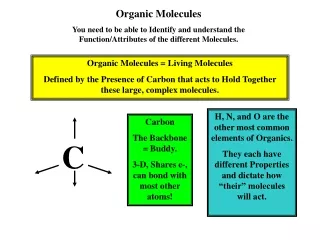
Complex Organic Molecules
E N D
Presentation Transcript
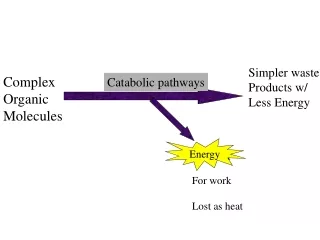
Simpler waste Products w/ Less Energy Complex Organic Molecules Catabolic pathways Energy ENERGY For work Lost as heat
“oxidation of glucose” ?????? Lose electrons = Oxidized (LEO) Gain electrons = Reduced (GER) Oxidation / Reduction reactions or Redox reactions have to occur in pairs Electrons move toward the more electronegative atoms Oxygen is most common electron acceptor
-NAD+ is a coenzyme -is found in all cells -helps transfer electrons
Overview of respiration -glucose is oxidized -electrons (hydrogen atoms) leave the carbon atoms and combine w/ O2 -this happens by a series of steps via NAD+ and an electron transport chain -During this process ATP is produced
Two ways to get ATP Oxidative phosphorylation 1 ATP production that is coupled to the exergonic transfer of electrons from food to oxygen
2 Substrate level phosphorylation ATP production by direct enzymatic transfer of phosphate from an intermediate substrate to ADP
Glycolysis overview -glucose (contain 6 Carbons) is split into two 3-carbon sugars. -these 3-carbon sugars are oxidized and rearranged to form 2 pyruvate molecules -occurs in the cytosol -no CO2 released -occurs whether or not oxygen is present. -2 net ATP produced
Figure 7.UN09 Oxidative phosphorylation: electron transport and chemiosmosis Citric acid cycle Pyruvate oxidation Glycolysis ATP ATP ATP
Figure 7.12 NADH 50 e− 2 NAD FADH2 Multiprotein complexes e− 2 FAD I 40 FMN II Fe•S Fe•S Q III Cyt b Fe•S 30 Cyt c1 IV Free energy (G) relative to O2 (kcal/mol) Cyt c Cyt a Cyt a3 20 e− 2 10 (originally from NADH or FADH2) 2 H ½ O2 0 H2O
Figure 7.14 2 1 H H Protein complex of electron carriers H H Cyt c IV Q III I ATP synthase II 2 H ½ O2 H2O FAD FADH2 NAD NADH ATP ADP P i (carrying electrons from food) H Electron transport chain Chemiosmosis Oxidative phosphorylation
Figure 7.13 H INTERMEMBRANE SPACE Stator Rotor Internal rod Catalytic knob ADP ATP P MITOCHONDRIAL MATRIX i
animation http://www.sumanasinc.com/webcontent/animations/content/atpsynthase.html
Figure 7.15 Electron shuttles span membrane MITOCHONDRION 2 NADH CYTOSOL or 2 FADH2 2 NADH 6 NADH 2 FADH2 2 NADH Oxidative phosphorylation: electron transport and chemiosmosis Glycolysis Pyruvate oxidation 2 Acetyl CoA Citric acid cycle 2 Pyruvate Glucose 2 ATP about 26 or 28 ATP 2 ATP About 30 or 32 ATP Maximum per glucose:
Brown Fat in hibernating animals Contain uncoupling protein that is a channel protein that allows diffusion of H+ NOT through ATP synthase
Comparison of chemiosmosis in chloroplasts and mitochondria SIMILARITIES An ETC in a membrane transports protons across a membrane ATP synthase in membrane couples diffusion of protons with phosphorylation of ADP ATP synthase and electron carriers (cytochromes) are very similar in both
DIFFERENCES ETC -mito transfer chemical E from food to ATP -electrons are extracted from oxidation of food molecules -chloroplasts transform light E into chemical E -uses light E to drive electrons to top of transport chain SPACIAL ORGANIZATION -mito pump protons from matrix out to the intermembrane space (which is a reservoir for protons) -chloro. Thylakoid membrane pumps protons from stroma into thylakoid compartment (serve as a proton reservoir)