Understanding the Classification of Matter: States, Properties, and Changes
This overview explores the classification of matter, detailing its various states: solids, liquids, gases, plasma, and Bose-Einstein condensates. It explains how matter is anything with mass and volume, emphasizing the unique characteristics of each state. The text distinguishes between physical and chemical properties, providing examples, and outlines physical and chemical changes. Furthermore, it differentiates between intensive and extensive properties, guiding the understanding of pure substances and mixtures. This foundational knowledge is essential for studying chemistry.

Understanding the Classification of Matter: States, Properties, and Changes
E N D
Presentation Transcript
Matter • Matter - the “stuff” – solids, liquids, gases, (plasma, and Bose-Einstein condensates) – that compose the universe • Matter is anything that has mass and takes up space
Molecules vibrate but can’t change position Retains shape and size SOLIDS
LIQUIDS • Atoms are close (similar to solids) – but can slip pass each other. • Has definite volume • No definite shape
GAS • Most energetic phase on Earth • Move fast – cannot attach to each other • No shape or volume
PLASMA • Results when gas is heated too a point where atoms lose electrons (10,000 C) • Stars • On Earth • Neon and Fluorescent lights (electrical charges passed through)
BOSE-EINSTEIN CONDENSATE • Created in 1995 • Occurs when temperature approach absolute zero (zero Kelvin) and all electrons exist at their lowest energy state. • Atoms do not appear as distinct particles but as one “super blob” Created 1995… Chemistry Jocks Predicted 1924...
Which of the following is not an example of matter? • Plasma • Wood • Your breath • Oxygen • A vacuum • water
What is matter composed of? • All matter is composed of atoms. • Atoms are the smallest particle of matter that still has the same properties of that type of element. (ex. copper, oxygen) • Multiple atoms chemically bonded together are called molecules (ex. H2or O2 oxygen gas)
If more than one type of atom (2 different capital letters) are bonded together, it is called a compound (ex. CuO – copper (II) oxide or MgS – magnesium sulfide) • (In other words, all compounds are molecules, but not all molecules are compounds.)
Classify the substance: CuCl • Atom • Molecule • Compound
Classify the substance: He • Atom • Molecule • Compound
Classify the substance: Cl2 • Atom • Molecule • Compound
Physical and Chemical Properties • A physical property describes the basic characteristics of a substance. • Examples: color, odor, taste, density, length, boiling point, melting point, volume, mass, temperature, phase (solid, liquid, or gas)
Chemical property • A chemical property describes how substances react with other substances. • Examples: wood burns in oxygen and gives off heat, iron rusts faster when exposed to oxygen and water
What kind of property?The boiling point of water is 373.15 Kelvin. • Physical property • Chemical property • Both • Neither
What kind of property?Platinum does not react with oxygen at room temperature. • Physical property • Chemical property • Both • Neither
What kind of property?Gallium metal melts in your hand. • Physical property • Chemical property • Both • Neither
What kind of property?Metal wire conducts electricity. • Physical property • Chemical property • Both • Neither
Physical and Chemical Changes • A physical change involves a change in physical properties (mass, temperature, phase change, etc.) but no change in the main components that make up the substance. • Ex. boiling, melting, breaking, slicing • A chemical change involves a change in the fundamental components of the substance and a new substance forms. • Ex. burning wood, iron rusting
Physical or Chemical Change?Iron metal is melted • Physical change • Chemical change • Both • Neither
Physical or Chemical Change?Milk turns sour • Physical change • Chemical change • Both • Neither
Physical or Chemical Change?A piece of wax is melted over fire and starts to burn. • Physical change • Chemical change • Both • Neither
Physical or Chemical Change?Steam from your shower condenses on the mirror. • Physical change • Chemical change • Both • Neither
Physical or Chemical Change?Electrolysis sends an electric current that splits water into hydrogen and oxygen. • Physical change • Chemical change • Both • Neither
Intensive & Extensive • Intensive Property: A property that does not depend on the amount of matter present • Extensive Property: A property that does depend on the amount of matter present
Organizational window We will not talk about any property that is both chemical and extensive.
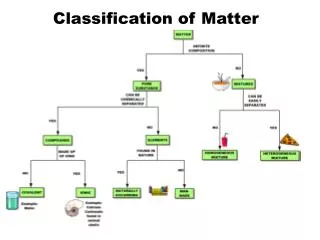
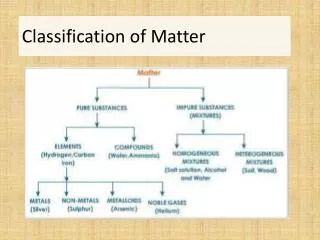
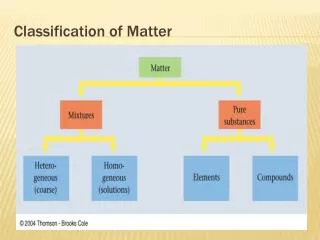
1 type of matter CAN NOT BE SEPARATED BY PHYSICAL MEANS 2 or more types of matter CAN BE SEPARATED BY PHYSICAL MEANS
Mixtures and Pure Substances • Pure substances always have the same composition, even at the molecular level. (ex. water, salt, nitrogen) • Impure substance of mixtures contain two or more materials that may be separated by physical means Examples of physical means include: filtration, distillation, chromatography, evaporation, sorting, magnetization
Heterogeneous Contains more than 1 type of matter Not uniform Homogeneous Contains more than 1 type of matter Same throughout MIXTURES Before it is Open? After it is Open? Examples: Chicken noodle soup Chocolate chip ice cream Examples: Soda pop Ink from a marker
1 type of matter CAN NOT BE SEPARATED BY PHYSICAL MEANS 2 or more types of matter CAN BE SEPARATED BY PHYSICAL MEANS
What type of matter?NaCl (table salt) • Mixture • Substance
What type of substance?NaCl (table salt) • Element • Molecule • Compound
What type of matter?Bowl of Lucky Charms (it’s magically delicious) • Element • Compound • Molecule • Homogeneous Mixture • Heterogeneous Mixture
Classify a pitcher of iced tea. • Element • Compound • Molecule • Homogeneous Mixture (solution) • Heterogeneous Mixture
Classify the air in the room. • Element • Compound • Molecule • Homogeneous Mixture (solution) • Heterogeneous Mixture
Classify oxygen gas (O2 ) • Element • Compound • Molecule • Homogeneous Mixture (solution) • Heterogeneous Mixture
Filtering • Separating out smaller and larger parts using a grating • Gravity Filtering: normal filtering where the drive to filter is solely derived by gravity pulling the smaller parts through the filter (Coffee or Tea Filtering) • Vacuum Filtering: filtering where a vacuum is used to pull the small particles through the filter (Büchner funnel)
Decanting (Pouring off) • Pouring off or removing one liquid without anything else • Naturally Separated • Centrifuge • Bottom Removal
Evaporative Separation • Separations based on changing one substance into its vapor state, then possibly recapturing it and returning it to a liquid • Evaporation: removal of a liquid by evaporation (Drying mud puddles) • Boiling: removal of a liquid by boiling it into a vapor (Reducing marsala sauce)
Evaporative Separation • Distillation: any process by which a liquid is removed through boiling or evaporation, then made to condensate and is captured as a liquid (Distillation of alcohol)
Pop Quiz! • Study physical separation… • Quiz will start at 2:11 and you will have 3 minutes to take it • Get a piece of paper out
1.) Name 2 physical properties • 2.) What is a molecule? • 3.) How would you separate alcohol from water?
Separations by Exotic Properties • Some methods of separation hinge upon the utilization of unusual properties, and are therefore rarely very useful • Magnetism • Sublimation
Separations by Exotic Properties • Chromatography – components of a mixture separate by dissolving and traveling at different rates through a mobile phase of chromatography paper