Proteins
Proteins. Digestion and absorption of proteins. The protein load received by the gut is derived from two primary sources: 70-100 g dietary protein, and 35-200 g endogenous protein,

Proteins
E N D
Presentation Transcript
Digestion and absorption of proteins • The protein load received by the gut is derived from two primary sources: 70-100 g dietary protein, and 35-200 g endogenous protein, • the latter either as secreted enzymes and proteins in the gut or from intestinal epithelial cell turnover • In healthy adults, only 1-2 g nitrogen, equivalent to 6-12g protein, are lost in the feces on a daily basis • Thus, the digestion and absorption of protein is extremely efficient
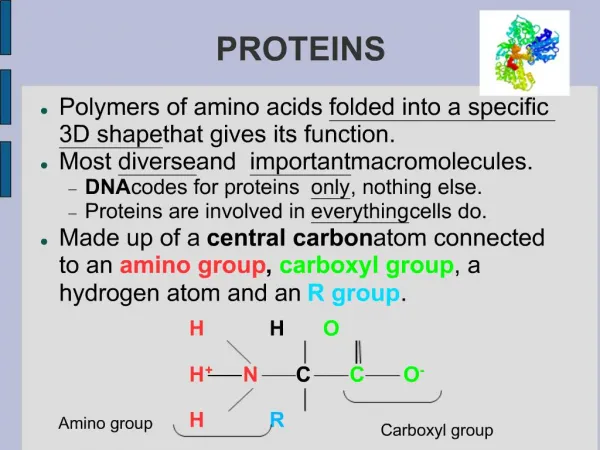
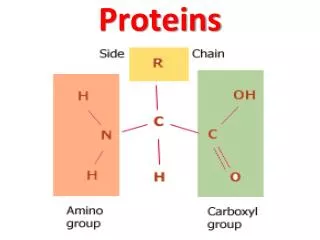
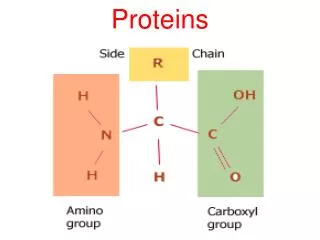
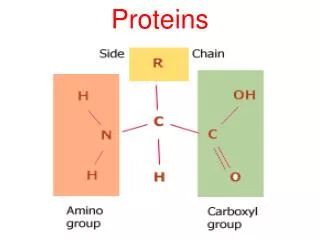
Peptidases hydrolyse proteins • Proteins, like other dietary macromolecules, are broken down by hydrolysis of specific peptide bonds and hence the enzymes involved are termed ‘peptidases’ • These enzymes can either cleave internal peptide bonds (i.e. endopeptidases) or cleave off one amino acid at a time from either the –COOH or –NH2 terminal of the polypeptide (i.e. they are exopeptidases subclassified into carboxypeptidases , and aminopeptidases, respectively)
The endopeptidases cleave the large polypeptides to smaller oligopeptides, which can be acted upon by the exopeptidases to produce the final products of protein digestion, amino acids, di- and tripeptides, which are then absorbed by the enterocytes • Depending on the source of the peptidases, the protein digestive process can be divided into gastric, pancreatic and intestinal phases
A. Digestion of proteins by gastric secretion The digestion of proteins begins in the stomach, which secretes gastric juice, a unique solution containing hydrochloric acid and the proenzyme pepsinogen: • Hydrochloric acid: Stomach acid is too dilute (pH 2-3) to hydrolyze proteins; however, the acid functions to kill some bacteria and to denature proteins, making them more susceptible to subsequent hydrolysis by proteases
Pepsin: This acid-stable endopeptidase is secreted by the serous cells of the stomach as an inactive zymogen (or proenzyme), pepsinogen In general, zymogens contain extra amino acids in their sequences, which prevent them from being catalytically active Note:Removal of these amino acids permits the proper folding required for an active enzyme
Pepsinogen is activated to pepsin either by HCL, or autocatalytically by other pepsin molecules that have already been activated • Pepsin releases peptides and a few free amino acids from dietary proteins
B. Digestion of proteins by pancreatic enzymes • On entering the small intestine, large polypeptides produced in the stomach by the action of pepsin are further cleaved to oligopeptides and amino acids by a group of pancreatic proteases • These enzymes, like pepsin, are synthesized and secreted as inactive zymogens
1. Release of zymogens: The release and activation of the pancreatic zymogens is mediated by the secretion of cholecystokinin and secretin, two polypeptide hormones of the digestive tract
2. Activation of zymogens: Enteropeptidase, an enzyme synthesized by and present on the luminal surface of intestinal mucosal cells of the brush border membrane, converts the pancreatic zymogen trypsinogen to trypsin by removal of a hexapeptide from the NH2-terminus of trypsinogen Trypsin subsequently converts other trypsinogen molecules to trypsin
Enteropeptidase thus unleashes a cascade of proteolytic activity, because • trypsin is the common activator of all the pancreatic zymogens
3. Specificity: Each of these enzymes has a different specificity for the amino acids R groups adjacent to the susceptible peptide bond
Products of pancreatic digestion • Thus, the products of digestion by pancreatic enzymes are: • Oligopeptides • Amino acids
Endopeptidases, dipeptidases and aminopeptidases complete the digestion of proteins • The final digestion of di- and oligopeptides is dependent on membrane-bound small intestinal endopeptidases, dipeptidases and aminopeptidases • The end products of this surface enzyme activity are free amino acids, di- and tripeptides
Absorption of amino acids di-, and tri-peptides • Free amino acids, di- and tripeptide are absorbed across the enterocyte membrane by specific carrier-mediated transport • Amino acids are transported by specific active transporters showing mechanisms which are similar to ones active in glucose transport
These Na dependant symporters are located at the brush- border membrane • This is an indirect active process • At least six specific symporter systems have been identified as follows: • Neutral amino acid symporter for a.a. with short or polar side- chains(Ser, Thr, Ala) • Neutral amino acid symporter for aromatic or hydrophobic side chains (Phe, Tyr,Tryp, Met,Val,Leu,Ileu) • Imino acid symporter (Pro, OH-Pro) • Basic amino acid symporter (Lys, Arg, Cys) • Acidic amino acid symporter ( Asp, Glu) • β - amino acid symporter ( β- Ala, Tau)
These transporter systems are also present in the renal tubules and • defects in their constituent protein structure can lead to disease( e.g. Hartnup disease) • Pathologies can be produced in both the kidney and intestine
Symporters for di-, and tri-peptides These are: • Present in the brush- border surface • H+- dependant
Di- and tripeptides are further hydrolyzed to their constituent amino acids inside the enterocyte • The final transfer is; therefore; of free amino acids across the contraluminal plasma membrane into the portal blood system • Na- independent transporters are present in the contraluminal surface, allowing a.a. facilitated transport to the portal vein
After a protein-rich meal, protein digestion takes place in the small intestine • The amino acids released are absorbed by intestinal epithelial cells • A large proportion of amino acids are transaminated to alanine, which is released into the portal vein and taken to the liver • Therefore, alanine is the major amino acid secreted by the gut and the principal carrier of nitrogen in the plasma
Defects in protein digestion and absorption • For a number of reasons, protein absorption might be incomplete • Some of the proteins, because of their physical or chemical structure, are resistant to proteolytic attack and therefore pass through the small intestine relatively unmodified • Furthermore, the absorption of free amino acids and peptides may be less than 100%, particularly if gut function is impaired
This occurs in a number of clinical conditions, such as intestinal infection or injury, and when certain ‘antinutritional’ factors such as lectins or trypsin inhibitor proteins are present in the diet • This unabsorbed protein or amino acid then passes through into the colon
Metabolism by the colonic microflora then occurs, but • the amino acids are no longer available to the body, and • are excreted in the faeces, mainly in the form of bacterial protein.
Abnormalities in protein digestion • In individuals with a deficiency in pancreatic secretion ,for example due to : chronic pancreatitis, cystic fibrosis, or surgical removal of the pancreas, • the digestion and absorption of fat and protein is incomplete • This results in the abnormal appearance of lipids, called “steatorrhea” , and undigested protein in the feces
Coeliac Disease • Characterized by severe mal-absorption and specific diagnostic features exhibited by the intestinal mucosa • The histological changes are due to the interaction of gluten, the principal protein of wheat, with the epithelium • The deficit is located within the mucosalcells, and permits certain polypeptides; resulting from digestion of gluten; to exert local effects within the intestine, and to be absorbed, inducing an antibody response
The intolerance is for gliadin portion of gluten ( found in wheat and several other grains)
Medical history • Diarrhea • Abdominal bloating • Weight loss Laboratory Features: • Anemia, Hgb of 9g/dl • Low serum iron, ferritin and Vit. B12 • Low RBC folate • Antibodies to gluten and its fractions are found in serum Histopathologigal features: • Biopsy demonstrated flattening of the mucosal surface, villous atrophy and disappearance of microvilli( reduction of surface, thus greatly reduced absorption
Nutritional Management • All gluten containing products should be removed from diet permanently(gluten-free diet) • Avoid wheat, barley, rye, and all products made with these grains • Oats may be tolerated, but this needs testing • Read labels carefully, because gluten containing grains are added to many products
Hartnup’s disease • A rare inherited disease of neutral amino acids symporter for aromatic or hydrophobic side chains (Phe, Tyr,Tryp, Met,Val,Leu,Ileu ) leading to malabsorption of these a.a.s • Produces a clinical syndrome mimicking pellagra (niacin deficiency- the three Ds) • Protein synthesis is normal • Treated by niacin supplements
Digestion of carbohydrate using a diagram if possible • Absorption of carbohydrate using a diagram if possible • The effect of food preparation on carbohydrate digestion and absorption
Colonic fermentation of carbohydrate to include types of carbohydrate reaching the colon • Physiological effects of carbohydrates not digested in the small intestine • Abnormal degradation of disaccharides • Modification of fats during food processing
Effect of heating on dietary oil • Digestion of dietary lipids using a diagram if possible • Absorption of dietary lipids using a diagram if possible • Hormonal control of lipid digestion
Defects if fat digestion and absorption including causes and the nutritional management of fat mal-assimilation • Digestion of dietary proteins using a diagram if possible • Absorption of dietary proteins using a diagram if possible
Defects in protein digestion and absorption • Coeliac disease • Steatorrhea (definition – causes – dietary management)