Mastering Acid Naming Techniques for Chemistry Success
90 likes | 119 Vues
Learn the formulas and naming conventions for binary, ternary acids in chemistry, including examples and key principles to grasp the concepts effectively.

Mastering Acid Naming Techniques for Chemistry Success
E N D
Presentation Transcript
Unit 7.3 Naming Acids Teacher: Dr. Van Der Sluys
Objectives • Formulas of acids • Naming acids
Formulas of Acids • An acid is a substance that when dissolved in water, dissociates or breaks apart into a hydrogen ion (H+) and an anion. • Acids always have hydrogen as the first element in the formula, • HCl (aq), HNO3 (aq), H2SO4 (aq), etc.
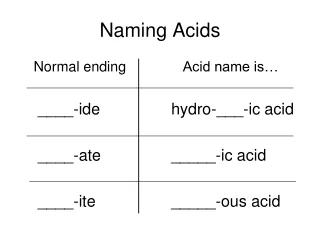
Naming Binary Acids • Binary acids contain only two elements, hydrogen and a monatomic anion whose name ends in the suffix -ide. • To name a binary acid like HCl • Begin the name with the prefix hydro- • Next use the root of the second elements name, e.g. chlor • Replace the suffix of the anion with the suffix -ic • Add the word acid • HCl is hydrochloric acid
Examples • HF • H2S • HBr • H2O • Hydroiodic acid • Hydronitric acid • Hydroselenic acid
Ternary Acids • Ternary acids contain three elements, with hydrogen being the first element in the formula and oxygen being the last. • To name a ternary acid such as H2SO4 • Remove the hydrogen ion(s) and determine the formula of the anion • Replace the suffix • -ate becomes -ic acid • -ite becomes -ous acid • H2SO4 anion SO42-sulfuric acid
Polyatomic Ions • Have you memorized the polyatomic ions yet?
Examples • HClO4 • H3PO4 • HNO3 • HNO2 • Acetic acid • Hypochlorous acid • Carbonic acid
Summary • Binary Acids Hydro ___ic acid • Ternary Acids If the anion is -ate it becomes _____ic acid If the anion is -ite it becomes _____ous acid