Oxidative Phosphorylation
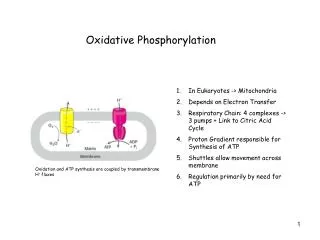
Oxidative Phosphorylation. Definition. It is the process whereby reducing equivalents produced during oxidative metabolism are used to reduce oxygen to water and produce ATP . It occurs in mitochondria , in eukaryotes.

Oxidative Phosphorylation
E N D
Presentation Transcript
Definition • It is the process whereby reducing equivalents produced during oxidative metabolism are used to reduce oxygen to water and produce ATP. • It occurs in mitochondria, in eukaryotes.
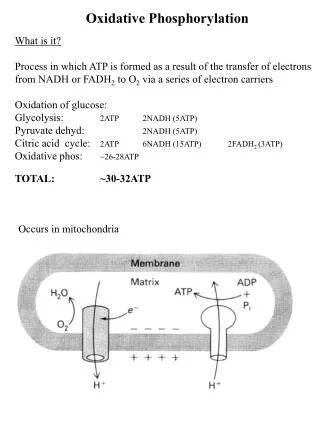
Reducing equivalents: NADH and FADH2 (nicotinamide adenine dinucleotide and flavin adenine dinucleotide). • Oxidative processes: • glycolysis • oxidative decarboxylation of pyruvate to acetyl CoA • TCA cycle, and • fatty acid oxidation
The reducing equivalents transfer their electrons through electron transport chain in inner membrane of mitochondria to O2, a terminal electron acceptor. • Releases large amount of free energy, which pumps H+ from the matrix across inner mitochondrial membrane, generating a transmembrane H+ gradient.
Energy from the H+ gradient drives the synthesis of ATP, catalyzed by the enzyme ATP synthase. • Oxidation (electron transport chain) is coupled to phosphorylation (ATP synthesis) by this transmembrane H+ fluxes. • This combined process of electron transport and ATP synthesis is called oxidative phosphorylation.
Biochemical anatomy of a mitochondrion
Mitochondria • Fig. 19-1 Lehninger • Has two membranes • Outer membrane - permeable to small molecules and ions • Inner membrane - impermeable to most small molecules and ions • Matrix - contains enzymes • Cristae - infoldings of the inner membrane
Electron transport chain • Electrons in NADH or FADH2 are transferred to oxygen through a series of reactions called the electron transport chain. • It is composed of electron transfer cofactors found in inner mitochondrial membrane.
Electron transfer cofactors • Ubiquinone (coenzyme Q) • Iron sulfur proteins • Cytochromes
Ubiquinone UQ (coenzyme Q) • Fig. 19-2 Lehninger • UQ - a hydrophobic compound with benzene ring, can diffuse within inner mitochondrial membrane. • UQ - can be reversibly reduced in two one-electron reductions forming the semiquinone radical and quinol.
Iron sulfur proteins • Fig. 19-5 Lehninger • Fe-S proteins : • contains iron bound to Cys in the protein. • iron undergoes reversible one-electron reduction from Fe3+ to Fe2+.
Cytochromes • Fig. 19-3 Lehninger • Complex organic molecule with Fe attached in the middle. • Iron undergoes reversible one-electron reduction from Fe3+ to Fe2+.
Electron transport chain (Respiratory chain) • The electron transport chain is organised into four multi-enzyme complexes: • complex I (NADH-Q reductase) • complex II (Succinate-Q reductase) • complex III (Cytochrome c reductase), and • complex IV (Cytochrome oxidase).
Separation of electron carriers • Fig. 19-7 Lehninger • The electron carriers of the respiratory chain are separated by gentle treatment of the inner mitochondrial membrane with detergents. • The resulting mixture is resolved by ion-exchange chromatography into different complexes.
Complex I - catalyzes electron transfer from NADH to ubiquinone • Complex II - catalyzes electron transfer from succinate to ubiquinone • Complex III - carries electrons from ubiquinone to cytochrome c • Complex IV - transfers electrons from cytochrome c to O2
Complex I(NADH-Q reductase) • Fig. 19-9 Lehninger • Mass: 850 kDa • no. of sub units: 42 • prosthetic groups: FMN, Fe-S • Overall reaction: • NADH + H+ + UQ -----> NAD+ + UQH2 • Oxidised UQ accepts hydride ion (H-) (two e- and one H+) from NADH and a H+ from the solvent water in the matrix.
Flow of electrons • NADH FMN Reduced Fe-S UQ • NAD+ FMNH2 Oxidised Fe-S UQH2 • NADH ---> FMN ---> Fe-S ---> UQ • Flow of 2e- from NADH to UQH2 leads to the pumping of 4H+ from matrix to cytosolic side of inner membrane.
Ubiquinol, UQH2 (reduced form) diffuses in the membrane from complex I to complex III where it is oxidised to ubiquinone, UQ. • Flow of e- from complex I ---> ubiquinone ---> complex III is accompanied by movement of protons from mitochondrial matrix to the outer side (cytosolic) of the inner membrane (the intermembrane space).
Complex II (succinate dehydrogenase) • Fig. 19-8 Lehninger • Mass : 140 kDa • No. of sub units : 5 • Prosthetic groups : FAD, Fe-S • Flow of electrons : • succinate ---> FADH2 ---> Fe-S ---> UQ
Succinate -----> Fumarate • FADH2 • FADH2 carries electrons from reactions by (i) glycerol phosphate dehydrogenase and (ii) fatty acyl CoA dehydrogenase and transfer it to UQ. • G0’ standard free energy change is too small, so no H+ is pumped out.
Complex III cytochrome c reductase • Fig. 19-10, 19-11 Lehninger • Mass : 250 kDa • No. of sub units : 11 • Prosthetic groups : heme, Fe-S • Flow of electrons : • UQ ---> cyt b ---> Fe-S ---> cyt c1 ---> cyt c • catalyses the transfer of electrons from UQ to cytochrome c.
Summary of flow of electrons • UQ cyt b UQH cyt c1 Fe-S cyt c (+2) (+3) • UQH cyt b UQH2 cyt c1 Fe-S cyt c (+3) (+2) • UQH2 + 2cyt c(Fe3+) + 2H+ ---> UQ + 2cyt c (Fe2+) + 4H+
Net effect: UQH2 is oxidised to UQ (H+ released) , cyt c is reduced, and movement of protons from matrix to intermembrane space (4H+) .
Complex IV, cytochrome oxidase • Fig. 19-13 Lehninger • Mass : 160 kDa • No. of sub units : 13 • Prosthetic groups : heme, CuA, CuB. • Flow of electrons: cyt a ---> cyt a3 ---> O2 • transfers electrons from cyt c to O2 to produce H2O.
Overall reaction: 4cyt c(Fe2+) + 8H+ + O2 ---> 4cyt c(Fe3+) + 4H+ + 2H2O
Summary of flow of electrons and protons through 4 complexes • Fig. 19-14 Lehninger • Complexes I & II : transfers e- to UQ • UQH2 passes e- to complex III • Complex III passes e- to cytochrome c • Complex IV transfers e- from reduced cytochrome c to O2 • e- flow through the complexes is accompanied by H+ flow from the matrix to intermembrane space.
Coupling of electron transport with ATP synthesis • Oxidative phosphorylation is a combination of two distinct activities: • (i) The flow of electrons from NADH (or FADH2) to oxygen via electron transport chain. NADH + H+ + 1/2O2 ---> NAD+ + H2O G’o = -nFE’o = -220kJ/mol • (ii) The phosphorylation of ADP to ATP. ADP + Pi ---> ATP + H2O G’o = +31kJ/mol
Flow of e- from NADH to O2 • Half-reaction (redox reaction) • NAD+ + H+ + 2e- ---> NADH, E’o = -0.32V • 1/2O2 + 2H+ + 2e- ---> H2O, E’o = +0.816V E’o = 1.14V • G’o=-2(96.5kJ/V.mol)(1.14V)=-220kJ/mol • Net reaction: exergonic • This energy is used to pump H+ out of the matrix
Phosphorylation of ADP to ATP • This reaction is catalyzed by an enzyme in the inner mitochondrial membrane, ATP synthase. • The large energy from reaction (i) is collected in the form of proton gradient.
Evidence for the coupling of e- transport and ATP synthesis • Fig. 19-17 Lehninger • Isolated mitochondria are suspended into buffered medium with an O2 electrode to monitor O2 consumption. At intervals, samples are assayed for the presence of ATP.
Addition of ... • ADP + Pi: no O2 consumption (no electron flow) and no ATP synthesis. • Succinate : e- transported to O2, H+ gradient is generated and ATP is synthesized. • CN- : no electron flow, no O2 consumption and no ATP synthesis. • Electron transfer and ATP synthesis are obligatorily coupled, i.e. neither reaction occurs without the other.
Addition of ... • Succinate : no electrons transported (obligatorily coupled), no ATP synthesis. • ADP + Pi : electrons transported to O2, and ATP synthesized. • Oligomycin : inhibitor of ATP synthase, no ATP synthesis and no e- transported to O2. • Dinitrophenol (DNP) : uncoupler, electrons transported to O2, no ATP synthesis.
Oligomycin : • inhibitor of ATP synthase, flow of protons into matrix is blocked • PMF builds up • when G for pumping H+ out of the matrix is > G relesed by the transfer of electrons from NADH to O2. • Electron flow stops and equilibrium is attained.
Fig. 19-18 Lehninger • DNP, dinitrophenol : • uncoupler, has a dissociable proton and is hydrophobic. • carries proton across inner mitochondrial membrane, dissipates the proton gradient
ATP synthesis • ATP synthesis is coupled to the dissipation of the proton gradient created during electron transport . • The proton flow back into the matrix through the proton pore in ATP synthase. • ADP + Pi + nH+ ---> ATP + H2O + nH+ intermembrane matrix
ATP synthase, complex V • ATP synthase is an F-type ATPase. • ATP synthase catalyzes the formation of ATP from ADP and Pi accompanied by the flow of protons from the intermembrane space to the matrix. • ATP synthase has two functional domains: F0 and F1.
Fo - integral membrane complex • sensitive to oligomycin • has a proton pore • F1 - peripheral membrane complex • essential for oxidative phosphorylation • binding site for ATP and ADP
Chemiosmotic model • Fig. 19-16 Lehninger • Proposed by Peter Mitchell (1960) • The chemiosmotic theory proposed that ATP synthesis is coupled to the dissipation of the proton gradient created during electron transport.
Synthesis of ATP depends on: • (i) Generation of proton gradient as the result of electron transfer through complexes I,III,& IV . • (ii) The inner membrane is impermeable to protons. • (iii) Protons move from the intermembrane space to the matrix space through a membrane pore in ATP synthase and this movement provides the driving force for ATP synthesis
(i) produces both chemical gradient, pH and electrical gradient, • (ii) protons reenter the matrix only through Fo. • PMF drives protons back into the matrix and provides the energy for ATP synthesis. • ATP synthesis is catalyzed by the F1 complex associated with Fo.