Oxidative Phosphorylation
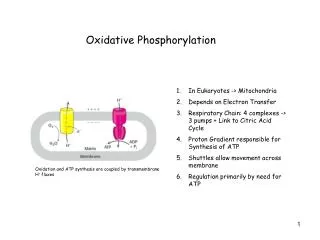
Oxidative Phosphorylation. In Eukaryotes -> Mitochondria Depends on Electron Transfer Respiratory Chain: 4 complexes -> 3 pumps + Link to Citric Acid Cycle Proton Gradient responsible for Synthesis of ATP Shuttles allow movement across membrane Regulation primarily by need for ATP.

Oxidative Phosphorylation
E N D
Presentation Transcript
Oxidative Phosphorylation • In Eukaryotes -> Mitochondria • Depends on Electron Transfer • Respiratory Chain: 4 complexes -> 3 pumps + Link to Citric Acid Cycle • Proton Gradient responsible for Synthesis of ATP • Shuttles allow movement across membrane • Regulation primarily by need for ATP Oxidation and ATP synthesis are coupled by transmembrane H+ fluxes
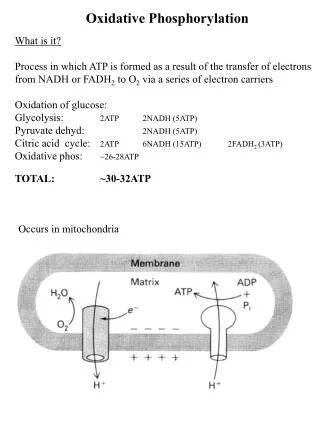
Oxidative Phosphorylation Oxidation of fuel (glucose, fat) -> formation of proton gradient -> drives synthesis of ATP
The Major Key Players in Oxidative Phosphorylation • ATP is the universal currency of free energy in biological systems • ATP -> ADP gives ΔGo’ = -7.3 kcal/mol • ATP->AMP gives ΔGo’ = -10.9 kcal/mol • ATP hydrolysis drives metabolism by shifting the equilibrium • Phosphoryl transfer potential is an important form of cellular energy transfer (Phosphorylated compounds are activated!!!)
The Major Key Players in Oxidative Phosphorylation Electron carrier for oxidation R = H -> NAD+ R = PO32- -> NADP+ !!! NAD+ accepts a H+ and 2 electrons (equivalent to a hydride ion H:-) -> NADH !!!
The Major Key Players in Oxidative Phosphorylation Electron carrier for oxidation FAD+ !!! FAD+ accepts 2 H+ and 2 electrons -> FADH2 !!!
Oxidative Phosphorylation takes place in the Inner Membrane of the Mitochondria
High Energy Electrons: Redox Potentials and Free-Energy Changes Electron transfer potential of NADH and FADH2 -> Phosphoryl transfer potential of ATP A 1.14 –Volt potential difference between NADH and O2 drives electron transport and favors formation of a proton gradient NADH 1.14 Volt O2
The Respiratory Chain Electron transfer from NADH -> O2 • Consists of 4 complexes: • 3 proton pumps + link to citric acid cycle • 3 proton pumps: • NADH-Q oxidoreductase • Q-cytochrome C oxidoreductase • Cytochrome c oxidase • Link to citric acid cycle: • Succinate-Q reductase • Ubiquinone (Coenzyme Q) also carries electrons from FADH2 (generated by citric acid cycle) generated through succinate-Q reductase Complex I Complex II -> Does not pump protons Ubiquinone Complex III Cytochrom c is an electron shuttle Complex IV
Electrons of NADH enter at NADH-Q Oxidoreductase • Enormous enzyme (>900 kDa) -> 46 polypeptides • proton pump • Steps of Electron-Transfer: • Binding of NADH and transfer of its electrons to FMN (prosthetic group of complex) • Electrons are transfered from FMNH2 to a series of iron-sulfur clusters (prosthetic group of complex) -> 2Fe-2S + 4Fe-4S clusters • Electrons are shuttled to coenzyme Q (ubiquinone) • 2 Electrons from NADH to Coenzyme Q -> pumping 4 H+ out of matrix of mitochondria NADH-Q Oxidoreducatase (Complex I)
Oxidation states of flavins Iron-sulfur clusters NADH-Q oxidoreductase
Oxidation state of Quinones (Coenzyme Q) The reduction of ubiquinone (Q) to ubiquinol (QH2) proceeds through a semiquinone intermediate (QH.)
Coupled Electron-Proton Transfer Reduction of Q -> QH2 results in uptake of 2 protons from matrix Coenzyme Q has the ability to transfer electrons -> used as an antioxidant (dietary supplement). CoQ10 used for the treatment of -> heart disease (especially heart failure), and also breast cancer Young people are able to make Q10 from the lower numbered ubiquinones such as Q6 or Q8. -> The sick and elderly may not be able to make enough, thus Q10 becomes a vitamin later in life. Supplementation of Coenzyme Q10 has been found to have a beneficial effect on the condition of some sufferers of migraine headaches. It is also being investigated as a treatment for cancer, and as relief from cancer treatment side effects. Some of these studies indicate that Coenzyme Q10 protects the brain from neurodegenerative disease such as Parkinsons and also from the damaging side effects of a transient ischemic attack (stroke) in the brain.
Ubiquinol is the Entry Point for Electrons from FADH2 of Flavoproteins FADH2 (citric acid cycle) • Complex II: • Integral membrane protein (inner mitochondrial membrane) • Electrons of FADH2 are transfered to Fe-S center and then to Q • No transport of protons
Ubiquinol is the Entry Point for Electrons from FADH2 of Flavoproteins FADH2 (citric acid cycle) • Succinate is oxidized to fumarate by the Succinate dehydrogenase A subunit. SDHA contains (FAD) cofactor The oxidized FAD -> reduced to FADH2 in a two electron process. This is part of the citric acid cycle. • The electron transfer subunit (SDHB) contains several iron-sulfur centers which relay electrons from SDHA to the membrane domains: a [2Fe-4S] cluster, a [4Fe-4S] cluster and a [3Fe-4S] cluster. • SDHC/SDHD dimer, reducing it to ubiquinol (QH2). • The resulting ubiquinol molecule is released, free to diffuse through the inner mitochondrial membrane to interact with subsequent enzymes of the mitochondrial respiratory chain (electron transport chain).
Electrons Flow from Ubiquinol (QH2) to Cytochrome c Through Q-Cytochrome c Oxidoreductase • Complex III: • Cytochrome is a electron - transfering protein • Cytochrome has a prosthetic group -> heme • Fe in heme group changes between +2 or +3 during e-transport • Function: catalyse transfer of electrons from QH2 -> oxidized cyt c • pumps protons out of matrix -> intermembrane space • Coupling of electron transport from Q -> cyt c and transmembrane proton transport Q cycle Heme group in cyt c
The Q Cycle Electrons that are bound to QH2 are transfered -> trigger uptake of 2 protons from the matrix -> formation of proton gradient 1st half of Q cycle 2nd half of Q cycle Q pool Q pool
Cytochrome c Oxidase Catalyzes the Reduction of O2 -> H2O • Complex IV: • Oxidation of cyt c coupled to reduction of O2 -> H2O • Heme protein • Heme + other part of active site (CuB) responsible for reduction of O2 • Electron transfer coupled to proton pump • 8 protons are pumped from the matrix to intermembrane space Transfer of 4 e- leads to safe product -> H2O
Reduction of O2 -> H2O can be toxic -> when single e- transfered In Peroxisomes -> catalase Superoxide dismutase deals with toxic derivates (superoxide radicals)
Mitochondrial electron transport chain -> drives ATP production
The Proton Gradient Powers Synthesis of ATP ATP sythesis mechanism
ATP Synthase is Composed of a Proton-Conducting Unit and a Catalytic Unit Proton gradient is not used to form ATP but to release ATP Proton channel Bind nucleotides – just β subunit catalysis synthesis (ATPase)
The World’s Smallest Molecular Motor -> Rotational Catalysis γ subunit rotates the 3 β-subunits driven by the proton-conducting unit ATP in tight (T) position -> cannot be released ATP in open (O) position -> released
The World’s Smallest Molecular Motor Fluorescently labeled actin filaments ATP hydrolysis -> counterclockwise rotation of filament (fluorescence microscope)
Proton Motion Across the Membran Drives Rotation of the C-Ring Each proton enters the cytosolic half-channel -> follows a complete rotation of the c-ring -> exits through the other half-channel into the matrix The difference in proton concentration and potential on the two sides -> leads to different probabilities of protonation through the 2 half-channels -> yields directional rotation motion c-ring
Shuttles between Mitochondria - Cytoplasma 1. Regeneration of NAD+ for glycolysis -> in respiratory chain (mitochondria) In Glycolysis -> cytoplasmic NAD+ -> cytoplasmic NADH Refill of NAD+ in cytosol
Shuttles between Mitochondria - Cytoplasma 1. Regeneration of NAD+ for glycolysis -> in respiratory chain (mitochondria) In Glycolysis -> cytoplasmic NAD+ -> cytoplasmic NADH need a shuttle to transfer -> cytoplasmic NADH into mitochondria (cannot just pass membrane) Transport of generated NADH into the mitochondria !!! and Refill of NAD+ in cytosol Just in the heart and liver cells !!!
Shuttles between Mitochondria - Cytoplasma 2. ATP/ADP transport by ATP/ADP translocase Oxidative phosphorylation generates ATP in the mitochondria -> needed in the cytoplasm need a shuttle to get -> cytoplasmic ADP into mitochondria (cannot just pass membrane)
Shuttles between Mitochondria - Cytoplasma Mitochondrial transporters (carriers)
Regulation of Respiration -> Primarily by Need for ATP ATPase inhibited by: Oligomycin and Dicyclohexylcarbodiimide (DCCD)
Regulated Uncoupling Leads to the Generation of Heat Uncoupling of oxidative phosphorylation -> heat generation to maintain body temperature Thermogenin UCP-1 (uncoupling protein) generates heat by short-circuiting the mitochondrial proton battery -> Special adipose tissue called ”Brown fat” (cells with high content of mitochondria , cytochrome -> brownish color) -> in these cells mainly heat generation (babies have a high amount of them)