OXIDATIVE PHOSPHORYLATION
OXIDATIVE PHOSPHORYLATION. ATP is the single currency of life. Adenosine triphosphate ATP is the most important molecule for capturing and transferring free energy

OXIDATIVE PHOSPHORYLATION
E N D
Presentation Transcript
ATP is the single currency of life • Adenosine triphosphate • ATP is the most important molecule for capturing and transferring free energy • Hydrolysis of ATP to ADP + Pi yields 7.3 kcal/mol energy that can be used to power e.g. protein synthesis, muscle contraction or transport of molecules
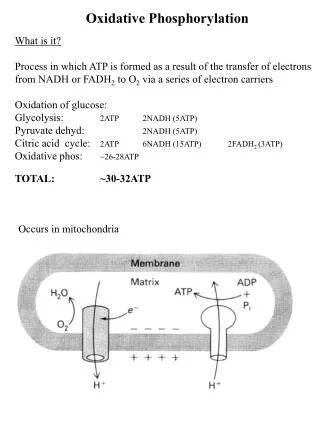
Oxidative phosphorylation generates ATP • In aerobic oxidation, sugars and fatty acids are metabolized to C02 and H20 . • The released energy is converted to chemical energy of phoshoanhydride bonds in ATP.
NADH and FADH2 • Glycolysis, TCA cycle and fatty acid oxidation generate NADH and FADH2 • NADH and FADH2 are energy rich molecules because each contains a pair of electrons that have a high transfer potential • In oxidative phosphorylation the electon transferring potentila of NADH and FADH2 is converted to phosphate-transfer potential of ATP
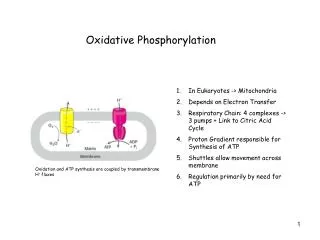
Oxidative phosphorylation • ATP is formed as electrons are transferred from NADH or FADH2 to 02 by a series of electron carriers.
Proton motive force and chemiosmotic coupling • The immediate energy sources that power ATP synthesis are proton gradient and electric potential (voltage gradient) across the membrane. • Proton gradient and electric potential are collectively called proton-motive force.
Proton motive force and chemiosmotic coupling • The proton motive force is generated by stepwise movement of electrons by electron carriers that leads to pumping of protons out of the mitochondrial matrix. • Oxidation of NADH and phosphorylation of ADP are coupled by a generation of proton gradient.
Energy is released gradually in the electron transfer chain • Most free energy released when glucose is oxidised to carbon dioxide is retained in the reduced coenzymes NADH and FADH2 • Respiration: electrons are released from from NADH and FADH2 to oxygen • NADH + H+ + 1/2 02 = NAD+ +H20
Energy is released gradually in the electron transfer chain • NADH + H+ + 1/2 02 = NAD+ +H20 -52.6 kcal/mol • ADP + Pi = ATP +7.6 kcal/mol • ATP production is maximised by releasing the free energy in small increments in the electron transfer chain (a.k.a respiratory chain). • Electron transfer chain contains four multiprotein complexes. Three of these are electron driven proton pumps that create the proton gradient
Redox potential • Oxidation-reduction potential • Oxidant + electron = reductant • Substance that can exist as a reduced and oxidices form is referred to a redox couple • Redox potential of such couple is measured against H+ -> H2 couple. • Redox potential of H+ -> H2 couple is defined as 0 V (volts).
Redox potential • A negative redox potential means that a substance has lower affinity for electrons than hydrogen. Positive redox potential means higher affinity. • Strong oxidising agents have positive redox potential • In the respiratory chain the electrons are transferred to higher redox potential values, that is, to higher affinity electron carriers.
Cytochromes are heme containing proteins • Cytochromes are covalently linked to heme, an iron-containing prosthetic group similar to that in hemoglobion or myoglobin. • Electron transport occurs by by oxidation and reduction of the Fe atom in the centre of the heme • Different cytochromes have slightly different heme groups that generate different ‘environment’ for Fe-ion and thus different tendency to accept an electron
ATP Synthase • ATP synthase or F0F1 complex has two components that are both itself multiprotein complexes • F0 is transmembrane complex that forms a regulated H+ channel • F1 is protrudes in the matrix and contains the sites for ATP formation
ATP Synthase • Proton translocation through F0 powers rotation of one subunits of F1 • Three confromations, one binds ADP and Pi so tightly that they spontaneously form ATP.
Malate/aspartate shuttle and glycerol phosphate shuttle are needed for oxidation of cytosolic NADH
Respiratory control • Mitochondria can only oxidise FADH and NADH only as long as there is ADP and Pi available. Electron flow ceases if ATP is not produced. • ADP increases when ATP is consumed e.g. in muscle work. Oxidative phosphorylation is regulated by ATP consumption.
Brown-fat mitochondria contain an uncoupler of oxidative phosphorylation • Brown fat specialised to produce heat • Newborns: brown-fat thermogenesis • Thermogenin protein, a proton transporter that is not connected to ATP synthesis. • Energy released by NADH oxidation converted to heat.