Oxidative Phosphorylation
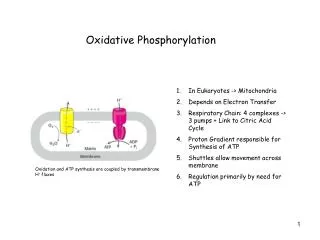
Molecular Biochemistry I. Oxidative Phosphorylation. Spontaneous e - flow through complexes I, III, & IV is coupled to H + ejection from the mitochondrial matrix. Stoichiometry: There is general agreement that

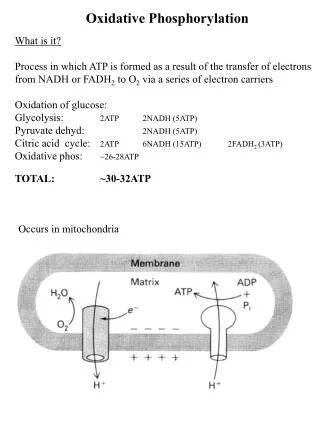
Oxidative Phosphorylation
E N D
Presentation Transcript
Molecular Biochemistry I Oxidative Phosphorylation
Spontaneous e- flow through complexes I, III, & IV is coupled to H+ejectionfrom the mitochondrial matrix. Stoichiometry: There is general agreement that • 4H+ are ejected from the mitochondrial matrix per 2e- passing through each of the complexes I and III. • The ratio is 2H+/2e- for complex IV.
Complex I (NADH Dehydrogenase): The mechanism of H+ transport linked to electron flow through complex I is still not known. There is evidence for conformational changes associated with transmembrane H+ transport, as with an ion pump.
Complex IV (Cyt. Oxidase): Protons are thought to pass through complex IV via a chain of groups subject to protonation/deprotonation, called a "proton wire." Proposed constituents of H+ transport pathways include: amino acid side-chains, propionate side-chains of the hemes, & water molecules within the complex.
There appears to be a separate pathway linking the buried binuclear center to each side of the membrane. A switch mechanism controlled by the reaction cycle is proposed to effect transfer of a H+ from one half-wire (half-channel) to the other. There cannot be an open pathway for H+ completely through the membrane, or oxidative phosphorylation would be uncoupled. The process of switching may involve conformational changes, and oxidation/ reduction-linked changes in pKa of groups associated with the metal centers.
Complex III (bc1 complex): H+ transport in complex III involves coenzyme Q (CoQ).
The “Q cycle” depends on: • mobility of CoQ in the lipid bilayer • existence of binding sites for CoQ within the complex that stabilize the semiquinone radical, Q·-.
Q Cycle: As depicted above, electrons enter complex III via coenzyme QH2, which binds at a site on the positive side of the inner mitochondrial membrane, adjacent to the intermembrane space.
QH2 gives up one e- that is transferred via hemes bL & bH to a bound Q on the other side of the membrane. Loss of one e- to the b hemes, and release of 2H+ to the intermembrane space, generates a Q·- radical in the site adjacent to the intermembrane space. Q·- becomes Q as it gives up a second e- to the Rieske iron-sulfur center (Fe-S).
Fe-S is reoxidized by electron transfer to cytochrome c1, which passes the electron out of the complex to cytochrome c. Some evidence suggests instead a concerted reaction in which e- transfer from QH2 to Fe-S & cytochrome bL is essentially simultaneous. But there is agreement about the overall reaction cycle.
It takes 2 cycles for CoQ bound at a site hear the matrix to be reduced to QH2, as 2e- are transferred from the b hemes, and 2H+ are extracted from the matrix compartment. In 2 cycles, 2QH2 enter the pathway & one is regenerated.
Animation Overall reaction catalyzed by complex III, including net inputs & outputs of the Q cycle : QH2 + 2H+(matrix) + 2 cyt c (Fe3+) Q + 4H+(outside) + 2 cyt c (Fe2+) Per 2e-transferred through the complex to cyt c, 4H+are released to the intermembrane space.
Complex III: Half of the homodimeric structure is shown. Approximate location of the membrane bilayer is indicated. Not shown are 2 CoQ binding sites, one near heme bH & the other near heme bL. The b hemes are positioned to provide a pathway for electrons across the membrane.
The Rieske iron-sulfur center (Fe-S) has a flexible link to the rest of the complex. It changes position during e- transfer. Fe-S extracts an e- from CoQ, & then moves closer to heme c1, to which it transfers the e-. (Fe-S protein in green.)
Complex III is an obligate homo-dimer. Fe-S in one half of the dimer interacts with bound CoQ & heme c1 in the other half of the dimer. Arrows point at: • Fe-S in the half of complex colored white/grey • heme c1 in the half of complex with proteins colored blue or green.
Simplified animation depicting: Ejection of a total of 20H+ from the matrix per 4e-transferred from 2NADH toO2 (10H+ per ½O2). Not shown is OH- that would accumulate in the matrix as protons, generated by dissociation of water (H2O H+ + OH-), are pumped out. Also not depicted is the effect of buffering.
A conventional view of mitochondrial structure is represented here. 3-D reconstructions based on electron micrographs of isolated mitochondria taken with a large depth of field, at different tilt angles have indicated that the infoldings of the inner mitochondrial membrane are actually variable in shape and are connected to the periphery and to each other by narrow tubular regions.
Electron micrograph by Dr. C. Mannella of a Neurospora mitochondrion in a frozen sample in the absence of fixatives or stains that might alter appearance of internal structures. See also Wadsworth Ctr. website. Apparently tubular cristae connect to the inner membrane via narrow passageways that may limit the rate of proton equilibration between the lumen of cristae & the intermembrane space.
ATP synthase, embedded in cristae of the inner mitochondrial membrane, includes: • F1catalytic subunit, made of 5 polypeptides with stoichiometry a3b3gde. • Fo complex of integral membrane proteins that mediates proton transport.
F1Fo couples ATP synthesis to H+ transport into the mitochondrial matrix. Transport of least 3 H+per ATP is required, as estimated from comparison of: • DG for ATP synthesis under cellular conditions (free energy required) • DGfortransfer of each H+ into the matrix, given the electrochemical H+ gradient (energy available per H+).
The Chemiosmotic Theory of oxidative phosphorylation, for which Peter Mitchell received the Nobel prize, states that coupling of ATP synthesis to respiration is indirect, via a H+ electrochemical gradient.
Chemiosmotic theory - respiration: Spontaneous e- transfer through complexes I, III, & IV is coupled to non-spontaneous H+ ejection from the matrix. H+ ejection creates a membrane potential (DY, negative in matrix) and a pH gradient (DpH, alkaline in matrix).
Chemiosmotic theory - F1Fo ATP synthase: Non-spontaneous ATP synthesis is coupled to spontaneous H+ transport into the matrix. The pH & electrical gradients created by respiration are the driving force for H+ uptake. H+ return to the matrix via Fo "uses up" pH & electrical gradients.
Transport of ATP, ADP, & Pi • ATP produced in the mitochondrial matrix must exit to the cytosol to be used by transport pumps, kinases, etc. • ADP & Pi arising from ATP hydrolysis in the cytosol must reenter the matrix to be converted again to ATP. • Two carrier proteins in the inner mitochondrial membrane are required. • The outer membrane is considered not a permeability barrier. Large outer membrane VDAC channels are assumed to allow passage of adenine nucleotides and Pi.
Adenine nucleotide translocase (ADP/ATP carrier) is an antiporter that catalyzes exchange of ADP for ATP across the inner mitochondrial membrane. At cell pH, ATP has 4 (-) charges, ADP 3 (-) charges. ADP3-/ATP4-exchange is driven by, and uses up, membrane potential (one charge per ATP).
Phosphatere-enters the matrix with H+ by an electroneutral symport mechanism. Pi entry is driven by, & uses up, the pH gradient (equivalent to one mol H+ per mol ATP). Thus the equivalent of one mol H+ enters the matrix with ADP/ATP exchange & Pi uptake. Assuming 3H+ transported by F1Fo, 4H+total enter the matrix per ATP synthesized. Animation
Questions: Based on the assumed number of H+ pumped out per site shown above, and assuming 4H+ are transferred back to the matrix per ATP synthesized: • What would be the predicted P/O ratio, the # of ATP synthesized per 2e- transferred from NADH to ½O2? • What would be the predicted P/O ratio, if the e- source is succinate rather than NADH?
Problem For, summing up synthesis of ~P bonds via oxidative phosphorylation, assume: 2.5 ~P bonds synthesized during oxidation of NADH produced via Pyruvate Dehydrogenase & Krebs Cycle (10 H+ pumped; 4 H+ used up per ATP). 1.5 ~P bonds synthesized per NADH produced in the cytosol in Glycolysis (electrons transferred via FAD to coenzyme Q). 1.5 ~P bonds synthesized during oxidation of FADH2 produced in Krebs Cycle (Succinate Dehydrogenase – electrons transferred to coenzyme Q).
An oxygen electrode may be used to record [O2] in a closed vessel. Electron transfer, e.g., NADH O2, is monitored by the rate of O2 disappearance. Above is represented an O2 electrode recording while mitochondria respire in the presence of Pi and an e- donor (succinate or a substrate of a reaction to generate NADH). The dependence of respiration rate on availability of ADP, the ATP Synthase substrate, is called respiratory control.
Respiratory control ratio is the ratio of slopes after and before ADP addition (b/a). P/O ratio is the moles of ADP divided by the moles of O consumed (based on c) while phosphorylating the ADP.
Chemiosmotic explanation of respiratory control: Electron transfer is obligatorily coupled to H+ ejection from the matrix. Whether this coupled reaction is spontaneous depends on pH and electrical gradients. ReactionDG e- transfer (NADHO2) negative value* H+ ejection from matrix positive; depends on H+ gradient** e- transfer with H+ ejection algebraic sum of above *DGo' = -nFDEo' = -218 kJ/mol for 2e- NADHO2. **For ejection of 1 H+ from the matrix: DG = RT ln ([H+]cytosol/[H+]matrix) + FDY DG = 2.3 RT (pHmatrix- pHcytosol) + FDY
With no ADP, H+ cannot flow through Fo. DpH & DY are maximal. As respiration/H+ pumping proceed, DG for H+ ejection increases, approaching that for e- transfer. When the coupled reaction is non-spontaneous, respiration stops. This is referred to as a static head. In fact there is usually a low rate of respiration in the absence of ADP, attributed to H+ leaks.
When ADP is added, H+ enters the matrix via Fo, as ATP is synthesized. This reduces DpH & DY. DG of H+ ejection decreases. The coupled reaction of electron transfer with H+ ejection becomes spontaneous. Respiration resumes or is stimulated.
Uncoupling reagents (uncouplers) are lipid-soluble weak acids. E.g., H+ can dissociate from the OH group of the uncoupler dinitrophenol. Uncouplers dissolve in the membrane and function as carriers for H+.
Uncouplers block oxidative phosphorylation by dissipating the H+ electrochemical gradient. Protons pumped out leak back into the mitochondrial matrix, preventing development of DpH or DY.
With uncoupler present, there is noDpH or DY. • DG for H+ ejection is zero • DG for e- transfer coupled to H+ ejection is maximal (spontaneous). Respiration proceeds in the presence of an uncoupler, whether or not ADP is present.
DG for H+ flux is zero in the absence of a H+ gradient. • Hydrolysis of ATP is spontaneous. The ATP Synthase reaction runs backward in presence of an uncoupler.
Uncoupling Protein An uncoupling protein (thermogenin) is produced in brown adipose tissue of newborn mammals and hibernating mammals. This protein of the inner mitochondrial membrane functions as a H+carrier. The uncoupling protein blocks development of a H+ electrochemical gradient, thereby stimulating respiration. DG of respiration is dissipated as heat. This "non-shivering thermogenesis" is costly in terms of respiratory energy unavailable for ATP synthesis, but provides valuable warming of the organism.