Renal Artery Stenosis
611 likes | 3.45k Vues
Renal Artery Stenosis. Resident’s conference Presented by: Gagandeep K Heer, MD (PGY-2). Background.

Renal Artery Stenosis
E N D
Presentation Transcript
Renal Artery Stenosis Resident’s conference Presented by: Gagandeep K Heer, MD (PGY-2)
Background • Renal artery stenosis (RAS) is the major cause of renovascular hypertension and it accounts for about 1-10% of the 50 million people in the United States who have hypertension. The incidence is less than1% of cases of mild to moderate HTN. However, it rises to 10 to 45 %in patients with acute (or superimposed upon a preexisting elevation in blood pressure), severe, or refractory hypertension. • Renovascular hypertension (RVHT) denotes nonessential hypertension in which a causal relationshipexists between anatomically evident arterial occlusive disease and elevated blood pressure. RVHT is the clinical consequence of renin-angiotensin-aldosterone activation as a result of renal ischemia. • RAS is also being increasingly recognized as an important cause of chronic renal insufficiency and end-stage renal disease. Studies suggest that ischemic nephropathy from RAS may be responsible for 5-22% of advanced renal disease in all patients older than 50 years in US.
Causes of RAS Major causes of the renal arterial lesions are: • Atherosclerosis —It is the cause of RAS in >2/3rd of the cases. This primarily affects men over the age of 45 and usually involves the aortic orifice or the proximal main renal artery. This disorder is particularly common in patients with diffuse atherosclerosis, but can occur as a relatively isolated renal lesion. • Fibromuscular dysplasia — In comparison to atherosclerosis, fibromuscular dysplasia most often affects younger women and typically involves the distal main renal artery or the intrarenal branches. • Other less common causes of RAS include: Vasculitis (Takayasu’s arteritis) Dissection of the renal artery. Thromboembolic disease Renal artery aneurysm Renal artery coarctation Extrinsic compression Radiation injury
Pathophysiology Atherosclerotic RAS • The initiator of endothelial injury is not clear; however, dyslipidemia, hypertension, cigarette smoking, diabetes mellitus, viral infection, immune injury, and increased homocysteine levels may contribute to endothelial injury. • In the atherosclerotic lesion site, endothelium permeability to plasma macromolecules (e.g. LDL) increases, turnover of endothelial cells and smooth muscle cells and intimal macrophages also increases. When atherogenic lipoproteins exceed certain critical levels, the mechanical forces may enhance lipoprotein insudation in these regions, leading to early atheromatous lesions. The sites where the initial atherosclerotic lesions developed are more prone to physiological adaptation to mechanical stress secondary to variations in flow and wall tension (e.g. renal artery site).
Pathophysiology Fibromuscular dysplasia: • Fibromuscular dysplasias are uncommon angiopathies associated with heterogeneous histologic changes that may affect the carotid circulation as well as the visceral and peripheral arteries. • String of beads is the classic radiographic finding seen in FMD. • FMD, as a cause of RAS usually affects young to middle-aged adults, mostly women, but it can also affect children.
Pathophysiology • Renal blood flow is 3 to 5 fold greater than the perfusion to other organs because it drives glomerular capillary filtration. Both glomerular capillary hydrostatic pressure and renal blood flow are important determinants of the glomerular filtration rate (GFR). • In patients with RAS, the GFR is dependent on angiotensin II and other modulators that maintain the autoregulation system between the afferent and efferent arteries and can fail to maintain the GFR when renal perfusion pressure drops below 70-85 mm Hg. Significant functional impairment of autoregulation, leading to a decrease in the GFR, is not likely to be observed until arterial luminal narrowing exceeds 50%. • RVHT develops as a result of increased renin and angiotensin II levels causing vasoconstriction as well as salt and water retention (volume expansion) due to increased aldosterone level.
Pathophysiology • Increased secretion of renin accelerates the conversion of angiotensin I to angiotensin II which enhances the adrenal release of aldosterone. • Angiotensin II causes vasoconstriction of both afferent and efferent arterioles, with a preferential affect on the efferent side. Under physiologic conditions, efferent tone is essential to maintain intraglomerular pressure. • In a kidney rendered ischemic by RAS with a reduced afferent blood flow, the intraglomerular pressure and glomerularfiltration are maintained by angiotensin II–mediated efferent vasoconstriction. Removal of the efferent vasoconstriction effect by using angiotensin blockade (e.g. by using ACEI) may reduce GFR by causing decrease in intraglumerular pressure. • In patients with RAS, the chronic ischemia produces adaptive changes in the kidney that are more pronounced in the tubular tissue. These changes include atrophy with decreased tubular cell size, patchy inflammation and fibrosis, tubulosclerosis, atrophy of the glomerular capillary tuft, thickening and duplication of the Bowman capsule, and intrarenal arterial medial thickening.
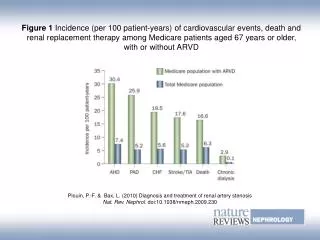
Demographics • Race: RVD is less common in African American patients. The incidence rate in 2 studies of patients with severe hypertension was 27-45% in whites compared to 8-19% in African Americans. • Sex: While the incidence of atherosclerotic RVD is independent of sex, Crowley et al showed that female sex (as well as older age, elevated serum creatinine level, coronary artery disease, peripheral vascular disease, hypertension, and cerebrovascular disease) is an independent predictor of RVD progression. • Age: The incidence of RAS increases with increasing age. In 1964, Holley et al reported data from 295 consecutive autopsies performed in their institution during a 10-month period. The mean age at death was 61 years. In the whole group, the prevalence rate of RAS was 27% of 256 cases identified as having history of hypertension. 56% of these showed significant stenosis (>50% luminal narrowing). Among normotensive patients, the incidence of severe RAS was 17%. Among those older than 70 years, 62% had severe RAS. Another study reported similar results, showing 18% incidence of severe RAS for patients aged 65-74 years and 42% for patients older than 75 years.
Clinical Findings • Difficult-to-control hypertension despite adequate medical treatment • Hypertension with renal failure or progressive renal insufficiency • Accelerated or malignant hypertension • Severe hypertension (diastolic blood pressure >120 mm Hg) or resistant hypertension • Hypertension with an asymmetric kidney • Paradoxical worsening of hypertension with diuretic therapy • Onset of hypertension occurring in patients younger than 30 years or older than 50 years • Symptoms of atherosclerotic disease elsewhere
Clinical Findings • Negative family history of hypertension • Cigarette smoking or use of other tobacco products • Renal failure with ACE inhibition. An increase in serum creatinine of more than 15% is strongly associated with a high incidence of RVD • Recurrent pulmonary edema (flash edema) • Advanced fundoscopic changes • Systolic-diastolic abdominal bruits (In combination with hypertension, these are suggestive of RVHT) • Unexplained renal insufficiency in elderly patients • Congestive heart failure with poor control of hypertension and renal insufficiency in the absence of a significant decrease in ejection fraction
Risk factors associated with ischemic renal disease • Carotid artery disease • Coronary artery disease • Diabetes mellitus • Hypertension • Obesity • Old age • Peripheral vascular disease (vascular disease in the extremities, e.g. the legs) • Smoking • Familial history of AD or RAS
Differential diagnosis • Acute renal failure • Chronic glomerulonephritis • Malignant HTN causing renal failure • Hypersensitivity nephropathy • Nephrosclerosis • Essential and other causes of hypertension with renal insufficiency
Mortality/Morbidity • In patients with hypertension, atherosclerotic renal artery disease is a strong predictor of increased mortality relative to the general population. In the setting of renal dysfunction, RVHT is associated with the greatest mortality rate. • Major complications of RVHT include end-organ damage due to chronically uncontrolled hypertension (CAD, stroke, etc.) and progressive renal insufficiency, which is an important sequel of chronic renal ischemia. • HTN may be particularly difficult to control or may require multiple antihypertensive agents (with increased adverse effects and drug interactions). • In addition, atherosclerotic RVD frequently occurs in the setting of generalized vascular disease (i.e. cerebral, cardiac, peripheral), with the consequences associated with disease in those vascular beds.
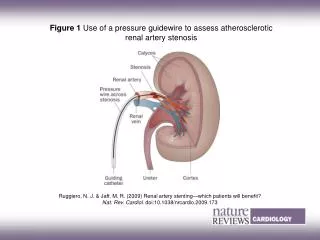
Work-up Laboratory Studies: • Serum creatinine and creatinine clearance. • 24-hour urine protein: Vascular renal disease is associated with minimal-to-moderate degrees of proteinuria, which are rarely in the nephrotic range. • Urinalysis shows absence of red blood cells or red blood cell casts (a hallmark of glomerulonephritis). • Serologic tests for SLE or vasculitis should be performed if these conditions are suggested (e.g. antinuclear antibodies, C3, C4, antinuclear cytoplasmic antibodies). • Measurement of plasma renin activity: The baseline plasma renin activity is elevated in 50-80% of patients with RVHT. • Captopril test: Measuring the increase in the baseline plasma renin activity 1 hour after the administration of 25-50 mg of the ACE inhibitor (captopril) can increase the predictive value of baseline plasma renin activity. Patients with RAS have an exaggerated increase in baseline plasma renin activity, perhaps due to the removal of the normal suppressive effect of high angiotensin II levels on renin secretion in the ischemic kidney. • Renal vein renin ratio ≥ 1.5 between stenotic/contralateral kidney are of considerable value in determining functionally important lesions and predicting cure or improvement of HTN with PTA or surgical intervention (although PTA or surgery will also benefit one-third to half the patients without lateralizing renal vein renin ratios).
Imaging Studies • Ultrasound/Duplex ultrasound: Renal US may show significant asymmetry of kidney size (i.e. size discrepancy of >1.5 cm). Additionally, US may be useful to determine the presence of a solitary kidney. Duplex ultrasound scanning is a noninvasive diagnostic technique that combines a B-mode ultrasound image with a pulse Doppler unit to obtain flow velocity data. It is noninvasive, relatively inexpensive, and can be used in patients with any level of renal function. • Captopril renography: Radionuclide renal imaging can be done using Tc99m DTPA, Tc99m MAG3 or OIH ( orthoiodohippuran). It is a safe and noninvasive way to evaluate renal blood flow and excretory function. When captopril is administered (especially in unilateral RAS), the GFR of stenotic kidney falls by about 30% and the normal kidney exhibits an increase in the GFR. Sensitivity of this test is about 85-90% and specificity of 93-98%. Significant azotemia and bilateral RAS adversely affect this accuracy, making it unsuitable for these situations. • CT angiography (Spiral CT): This technique involves the use of IV iodinated contrast material and allows 3-dimensional reconstruction images of the renal arteries. Spiral CT is a useful technique that avoids arterial catheterization and produces accurate images of renal artery anatomy.
Imaging Studies • Magnetic resonance angiography: Magnetic resonance angiography (MRA) is a very good noninvasive technique capable of demonstrating the renal vascular anatomy and direct visualization of renal artery lesions without iodinated contrast material. • The limitations of MRA are its expense and its contraindication in patients with metallic clips, pacemakers, intraocular metallic devices, or other implants. • The sensitivity of MRA > 90% for proximal RAS, 82% for main RAS, and very low ( 0%) for segmental stenosis.
Dynamic gadolinium-enhanced magnetic resonance angiogram (MRA) shows normal renal arteries.
Imaging • Renal arteriography: This technique remains the gold standard for the confirmation and identification of renal artery occlusion in persons with IRD. Specialists can perform renal arteriography by conventional aortography, intravenous subtraction angiography, intra-arterial digital subtraction angiography (DSA), or carbon dioxide angiography. • Conventional aortography produces excellent radiographic images of the renal artery. • It is, however, an invasive procedure that requires an arterial puncture, carries the risk of cholesterol emboli, and uses a moderate amount of contrast material with the risk of contrast-induced acute tubular necrosis (ATN). • Low osmolar contrast material can limit the risk of CEN.
Digital subtraction flush aortogram in a 77-year-old normotensive man shows marked left renal artery stenosis and diffuse aortic atheroma. The patient presented with lower-limb claudication.
Digital subtraction flush aortogram in an 83-year-old mildly hypertensive man shows complete occlusion of the left renal artery; only a stub of the artery is visualized. Note the diffuse aortic atheroma. The patient presented with lower-limb claudication.
Aortogram of a 4-year-old child with renovascular hypertension caused by stenosis of the left renal artery.
Flush aortogram in a 32-year-old man with familial hypercholesterolemia and difficult-to-control hypertension. Radiograph shows a complete occlusion of the right renal artery and marked stenosis of the left renal artery.
Left: Flush aortogram in a 63-year-old man with hypertension shows marked stenosis of the right renal artery and complete occlusion of the left renal artery. Note the extensive atheroma in the aorta and iliac arteries. • Right: nephrogram-phase image shows a significantly smaller left kidney with a faint nephrogram.
Selection of diagnostic tests • Once patients are identified as being at higher risk of RAS, the choice of the best test for diagnosis is controversial. • Accurate identification of patients with correctable renovascular hypertension can be difficult with use of standard noninvasive techniques (e.g. sonography, CT angio, MRA) because they provide only indirect evidence of the presence of renal artery lesions. • On the other hand, invasive techniques with more accurate diagnostic potential can produce a worsening of renal function because of contrast toxicity and complications related to the procedures themselves (e.g. arterial puncture, catheter-induced atheroembolism).
Selection of diagnostic tests • When the history is highly suggestive and no risk for radiocontrast-mediated renal injury is present, an intraarterial DSA or conventional angiogram is the appropriate initial test. In patients at risk, a carbon dioxide angiogram can determine the presence of a stenosis, and the risk of radiocontrast angiogram is imposed only on those individuals are most likely to benefit. • Perform a spiral CT scan, MRA, or duplex ultrasonography (depending on availability and local experience) when moderate suspicion of renovascular disease exists. A negative test result indicates that an RAS is highly unlikely, while a positive test result can be followed by renal arteriography.
Selection of Diagnostic Tests Gilfeather et al performed a study evaluating conventional angiography versus gadolinium-enhanced MRA in 54 patients and 107 kidneys. The study showed that in 70 kidneys (65%), the average degree of stenosis reported by readers of both modalities differed by 10% or less. In 22 cases (21%), MRA overestimated the degree of stenosis by more than 10% relative to the results of conventional angiography; in 15 cases (14%), MRA underestimated the stenosis by more than 10%.
Selection of Diagnostic Tests • The obvious advantages of conventional angiography are its ability to accurately determine the extent of the lesion and suggesting its clinical importance (by demonstrating post-stenotic dilation) and the ability to concurrently perform endovascular therapy. • However, specialists should weigh these advantages against the higher cost and greater morbidity of conventional angiography. • All these tests should be done if the pt is a reasonable candidate for revascularization procedure.
Prognosis • Images from sequential abdominal aortographs or duplex ultrasound scans in patients with documented renal artery lesions who have been treated medically have shown that progressive arterial obstruction occurs in 42-53% of patients with atherosclerotic RAS, often within the first 2 years of radiographic follow-up. • The incidence rate of progression to complete renal artery occlusion in these studies ranges from 9-16%;this often occurs in patients with a high-degree stenosis. In a study of 85 patients at the Cleveland Clinic who were followed for 3-172 months, patients with mild-to-moderate stenosis remained unchanged upon follow-up, and 39% of patients with greater than 75% lesions progressed to total occlusion. • The prognosis of patients with RVHT is difficult to ascertain and varies with the extent of the occlusive phenomena, the sensitivity of the individual to antihypertensive therapy, and the efficacy of surgical repair and/or angioplasty. • RVHT in the setting of chronic renal ischemia and consequent renal dysfunction has been linked to worse outcomes.
Treatment • It is crucial to remember that not all HTN in the presence of anatomic RAS is renovascular HTN. • Many normotensive patients and those with essential HTN can also have some degree of RAS and do not need revascularization.
Treatment Interventions appropriate for patients with RAS/RVHT may include • Medical therapy • Percutaneous transluminal angioplasty (PTA) with or without vascular stent placement • Surgical revascularization • Intravascular ultrasonography-guided atherectomy
Medical therapy • Treatment with antihypertensive drugs is indicated and optimal blood pressure control is essential. ACE inhibitors should be avoided. CCB, beta blockers and many other classes of drugs can be safely used. In many patients, blood pressure can be well controlled with medical therapy. • Other risk factors that should be addressed include atherosclerosis, smoking, and hyperlipidemia. • Definitive therapy for the RAS should always be considered not only for better control of HTN but also to prevent the development of ischemic nephropathy. • In patients with diffuse atherosclerosis, the complication rate with both surgery and angioplasty is relatively high. Medical therapy may be preferred to other treatments many cases after carefully weighing risks and benefits of the invasive intervention.
Percutaneous transluminal angioplasty (PTA) • PTA has become the procedure of choice for treatment of symptomatic stenoses. Patency rates after PTA are strongly dependent on the size of the vessel treated and the quality of inflow and outflow through the vessel. Because of the excellent results obtained with renal angioplasty, it is the most commonly performed procedure in symptomatic RAS. • Previously, a solitary or transplanted kidney was considered a contraindication for PTA. This is no longer the case and, angioplasty is now considered the procedure of choice for treatment of RAS in these patients.
PTA • Technical success is achieved in more than 90% of patients, and patency rates are 90-95% at 2 years for FMD and 80-85% for atherosclerosis. • Restenosis requiring repeat angioplasty has been reported in fewer than 10% of patients with FMD and in 8-30% with atherosclerotic stenosis. • Improvement in blood pressure control with fewer antihypertensive medications is achieved in 30-35% of fibromuscular lesions and in 50-60% of atherosclerotic lesions. • A success rate of 83% has been reported with PTA in RAS associated with renal transplantation.
Left: A balloon angioplasty catheter is seen in situ across the left renal artery stenosis. • Right: After angioplasty, an excellent anatomic (and functional) result was achieved.
Left renal artery stenosis After PTA
Vascular stent placement • Vascular stenting is considered complementary to PTA. • Many vascular stents are now available, which can be either self-expanding or balloon expandable. Drug-eluting stents are also available. • Intravascular stents placed during angioplasty may be helpful in the prevention of restenosis. • Early results suggest that stenting may prove useful in patients with ostial disease, in those in whom restenosis occurs after PTA, or in those with complications (e.g. renal artery dissection) resulting from PTA. • Primary renal artery stenting in patients with atherosclerotic RAS has a high technical success rate and a low complication rate.
Intravascular ultrasonography-guided atherectomy • In a single reported case, hypertension secondary to AD was successfully diagnosed with intravascular sonography, and intravascular sonography-guided renal atherectomy was curative.
Surgical revascularization • Currently, surgical revascularization is reserved for patients in whom the main renal artery appears completely occluded and in whom the surviving renal parenchyma is vascularized by collaterals. • Surgical revascularization might also be used when an ostial stenosis is present with a buttressing atheroma on either side of the ostium.
Surgery • Several surgical options are available. • The stenotic segment may be excised and the artery resutured directly onto either the aorta or surviving stump. • A vein graft may be transplanted or the kidney resected and reimplanted in the iliac fossa with the renal artery anastomosed to the iliac artery. • Another novel method involves a splenectomy and anastomoses of the splenic artery to the renal artery when RAS involves the left kidney. The underlying diagnosis determines the results of this surgery. • With advanced diffuse atherosclerosis, surgery may become less feasible because the certainty that the RAS is the cause of the hypertension is less and the prognosis may be determined by comorbidities.
Surgery • A potential complication of surgery is the release of cholesterol emboli during the surgery which can cause renal failure; however, 80-90% of patients undergoing operation for atherosclerotic RAS benefit with cure or improvement. The perioperative mortality rate is less than 5%. • In patients with FMD, the cure rate is as high as 80%, and morbidity rates are low. However, these results are not significantly better than what can be achieved with renal angioplasty, at less morbidity, mortality, cost, and inconvenience. • In patients with diffuse atherosclerosis, the complication rate with both surgery and angioplasty is relatively high.
Criteria for Revascularization • All patients with bilateral stenosis and stenosis in a solitary functioning kidney are candidates for revascularization, regardless of whether they have renal insufficiency. • When renal insufficiency is present, patients with unilateral stenosis are also possible candidates for revascularization
Criteria for Revascularization • When renal function is normal or nearly normal, specialists recommend revascularization if the patient meets the following criteria: • The degree of stenosis is more than 80-85%. • The degree of stenosis is 50-80%, and captopril-enhanced scintigraphy findings demonstrate an activation of intrarenal renin angiotensin system. • Conversely, physicians can choose observation instead of revascularization (serial imaging every 6 mo with duplex scanning, accurate correction of dyslipidemia, use of drugs that block platelet aggregation) when the patient meets the following criteria: • Stenosis is 50-80%, and scintigraphy findings are negative. • The degree of stenosis is less than 50%.
Criteria for Revascularization • When renal insufficiency is present and the objective is recovery of renal function together with prevention of further renal function impairment, the prerequisites for revascularization are as follows: • The serum creatinine level is lower than 4 mg/dL. • The serum creatinine level is higher than 4 mg/dL but with a possible recent renal artery thrombosis. • When these conditions are satisfied, revascularization can be done if the following apply: • The degree of stenosis is more than 80%. • The serum creatinine level is increased after administration of ACE inhibitors. • The degree of stenosis is 50-80%, and the scintigraphy findings are positive.
When to chose conservative therapy • Restrict conservative treatment in patients with an established diagnosis of IRD only to those with absolute contraindications to surgery or angioplasty or to patients who are likely to succumb due to other comorbid conditions before advancing to end-stage renal disease because of IRD. • In these cases, the clinicians have to rely on pharmacologic agents (e.g. combination of calcium channels blockers to control blood pressure and optimize renal perfusion), accepting the high probability of deterioration in renal function.