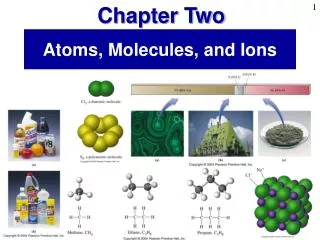
Atoms, Molecules and Ions
1.18k likes | 2.02k Vues
Atoms, Molecules and Ions. Atoms. Each element composed of atoms All atoms of a given element are identical Atoms of an element are not changed during a chemical reaction (the atoms just move from one chemical species to another) Compounds are formed when atoms of more than 1 element combine.

Atoms, Molecules and Ions
E N D
Presentation Transcript
Atoms • Each element composed of atoms • All atoms of a given element are identical • Atoms of an element are not changed during a chemical reaction (the atoms just move from one chemical species to another) • Compounds are formed when atoms of more than 1 element combine
Atoms • John Dalton • The Atomic Theory of Matter • Credited with developing Natural Laws
Atoms • Law of conservation of mass • Law of constant composition • Law of multiple proportions: When two elements form different compounds, the mass ratio of the elements in one compound is related to the mass ratio in the other by a small whole number.
Discovery and Properties of Electrons • Humphrey Davy (early 1800’s) - passed electricity through compounds • compounds decomposed into elements • compounds are held together by electrical forces • Michael Faraday - (1832-1833) - amount of reaction that occurs during electrolysis is proportional to current passed through compounds • Matter (atoms) is electrical in nature.
Discovery and Properties of Electrons • Cathode Ray Tubes - (late 1800’s & early 1900’s) • 2 electrodes in a glass tube with a gas at low pressure • voltage applied to tube causing a glow discharge • “rays” emitted from cathode (- end) to anode (+ end) • Cathode Rays must be negatively charged!
Discovery and Properties of Electrons • J.J. Thomson - (1897) - changed cathode ray tube experiments by adding two adjustable voltage electrodes into the experiment
Discovery and Properties of Electrons • measured charge to mass ratio of electrons • e/m = -1.75881 x 108 coulomb/g of e- • named cathode rays electrons • Thomson is the “discoverer of electrons”
Discovery and Properties of Electrons • Robert A. Millikan - 1st American Nobel Laureate • determined the charge and mass of the electron (1909) • oil drop experiment
Discovery and Properties of Electrons • charge on 1 electron = -1.60219 x 10-19 coulomb • using Thomson’s charge to mass ratio we get that the mass of 1 electron is 9.11 x 10-28 g
Canal Rays and Protons • Goldstein (1886) - “Canal Rays” • streams of positively charged particles in cathode rays • flow in opposite direction of cathode rays • must be positive • postulated existence of “proton”
Discovery of the Nuclear Atom • Plum-Pudding? • Thomson assumed all these charged species were found in a sphere.
Rutherford and the Nuclear Atom • Ernest Rutherford - 1910 - basic picture of atom Geiger & Marsden’s experiment on a- particle scattering from thin Au foils
Rutherford and the Nuclear Atom • In order to get the majority of -particles through a piece of foil to be undeflected, the majority of the atom must consist of a low mass, diffuse negative charge - the electron. • To account for the small number of high deflections of the -particles, the center or nucleus of the atom must consist of a dense positive charge.
Rutherford and the Nuclear Atom • Rutherford decoded the scattering information
Rutherford and the Nuclear Atom • atom is mostly empty space • very small, dense center called nucleus • nearly all of atom’s mass in nucleus • nuclear diameter is 1/10,000 to 1/100,000 times less than atom’s radius • nuclear density is 1015g/mL • equivalent to 3.72 x 109 tons/in3
Neutrons • James Chadwick - 1932 • analyzed evidence from a-particle scattering off Be • recognized existence of massive neutral particles - “neutrons”
The Modern View of Atomic Structure • The atom consists of positive, negative, and neutral entities (protons, electrons, and neutrons). • Protons and neutrons are located in the nucleus of the atom, which is small. Most of the mass of the atom is due to the nucleus. • There can be a variable number of neutrons for the same number of protons. Isotopes have the same number of protons but different numbers of neutrons. • Electrons are located outside of the nucleus. Most of the volume of the atom is due to electrons.
Fundamental Particles • Three fundamental particles make up atoms. The following table lists these particles together with their masses and their charges.
Mass Number & Isotopes • H.G. J. Moseley (1912-1914) - recognized that atomic number is the defining difference between elements • new understanding of Mendeleev’s periodic law
Atomic Number • Sometimes given the symbol Z • number of protons in the nucleus • determines the element • also determines number of electrons in a neutral atom
Isotopes, Atomic Numbers & Mass Numbers • All atoms of an element have the same number of protons in the nucleus • Isotopes of an atom have a different number of neutrons in the nucleus • Atomic number = # of protons • Mass number = # protons + # neutrons
Isotopes, Atomic Numbers & Mass Numbers • By convention, for element X, we write • Isotopes have the same Z but different A.
Isotopes • Give the number of protons, neutrons and electrons in each of the following species: 56Fe 56Fe3+31P 31P3-
Isotopes • Give the number of protons, neutrons and electrons in each of the following species: 56Fe 56Fe3+35Cl-131P3- P 26 N 30 E 26
Isotopes • Give the number of protons, neutrons and electrons in each of the following species: 56Fe 56Fe3+35Cl-131P3- P 26 26 N 30 30 E 26 23
Isotopes • Give the number of protons, neutrons and electrons in each of the following species: 56Fe 56Fe3+35Cl-131P3- P 26 26 17 N 30 30 18 E 26 23 18
Isotopes • Give the number of protons, neutrons and electrons in each of the following species: 56Fe 56Fe3+35Cl-131P3- P 26 26 17 15 N 30 30 18 16 E 26 23 18 18
Atomic Weights • weighted average of the masses of the constituent isotopes • lower number on periodic chart
The Periodic Table • The Periodic Table is used to organize the 114 elements in a meaningful way. • As a consequence of this organization, there are periodic properties associated with the periodic table.
The Periodic Table • Columns in the periodic table are called groups (numbered from 1A to 8A or 1 to 18). • Rows in the periodic table are called periods. .
The Periodic Table • Metals are located on the left hand side of the periodic table (most of the elements are metals). • Non-metals are located in the top right hand side of the periodic table. • Elements with properties similar to both metals and non-metals are called metalloids and are located at the interface between the metals and non-metals.
The Periodic Table • Some of the groups in the periodic table are given special names. • These names indicate the similarities between group members: Group 1A: Alkali metals. Group 2A: Alkaline earth metals. Group 6A: Chalcogens. Group 7A: Halogens. Group 8A: Noble gases.
The Periodic Table • Name the following elements. Indicate if each is a metal, nonmetal, or metalloid. • P Sn Mn • K Cu Hg • F As N • Si Na Ca • Fe Ag Mg
The Periodic Table • Name the following elements. Indicate if each is a metal, nonmetal, or metalloid. • P phosphorusSntinMnmanganese • KPotassiumCucopperHgmercury • F fluorineAsarsenic N nitrogen • SisiliconNasodiumCacalcium • FeironAgsilverMg magnesium
Molecules and Molecular Compounds • Molecules are assemblies of two or more atoms bonded together. • Each molecule has a chemical formula. • The chemical formula indicates • which atoms are found in the molecule • in what proportion they are found. • Compounds formed from molecules are molecular compounds.
Molecules and Molecular Compounds • empirical formula - simplest molecular formula, shows ratios of elements but not actual numbers of elements • molecular formula - actual numbers of atoms of each element in the compound
Molecules and Molecular Compounds • Molecules occupy three dimensional space. • However, we often represent them in two dimensions. • The structural formula gives the connectivity between individual atoms in the molecule. • The structural formula may or may not be used to show the three dimensional shape of the molecule.
Molecules and Molecular Compounds • If the structural formula does show the shape of the molecule, then either a perspective drawing, ball-and-stick model, or space-filling model is used.
Chemical Formulas • show the ratio of the elements present in the molecule or compound • He, Au, Na - monatomic • O2, H2, Cl2 - diatomic • O3, S8, P4 - more complex elements • H2O, C12H22O11 - compounds
Ions & Ionic Compounds • ions are atoms or groups of atoms that are charged • two basic types of ions • positive ions or cations • one or more electrons less than neutral • negative ions or anions • one or more electrons more than neutral
Ions and Ionic Compounds • The number of electrons an atom loses is related to its position on the periodic table. • Metals tend to form cations whereas non-metals tend to form anions.
Ions and Ionic Compounds • The majority of chemistry involves the transfer of electrons between species. (Ionic Bonding) • Example: • To form NaCl, the neutral sodium atom, Na, must lose an electron to become a cation: Na+. • The electron cannot be lost entirely, so it is transferred to a chlorine atom, Cl, which then becomes an anion: Cl-. • The Na+ and Cl- ions are attracted to form an ionic NaCl lattice which crystallizes.
Ions & Ionic Compounds • Sodium chloride - table salt is an ionic compound
Ion Names and Formulas • Common Polyatomic Ions • Can be found on my website • Can be found as a table in your book • LEARN THEM!!!!