Polymer Synthesis and Hydrolysis: Reactions, Structures, and Functions
Explore the processes of polymer synthesis and hydrolysis, including dehydration reactions and breaking down polymers. Learn about the different structures and functions of carbohydrates, proteins, and fats. Includes images and examples.

Polymer Synthesis and Hydrolysis: Reactions, Structures, and Functions
E N D
Presentation Transcript
(a) Dehydration reaction: synthesizing a polymer 1 2 3 Short polymer Unlinked monomer Dehydration removesa water molecule,forming a new bond. Figure 5.2 1 4 2 3 Longer polymer (b) Hydrolysis: breaking down a polymer 4 2 3 1 Hydrolysis addsa water molecule,breaking a bond. 2 3 1
(a) Dehydration reaction: synthesizing a polymer 2 1 3 Unlinked monomer Short polymer Figure 5.2a Dehydration removesa water molecule,forming a new bond. 4 2 1 3 Longer polymer
(b) Hydrolysis: breaking down a polymer 4 2 1 3 Figure 5.2b Hydrolysis addsa water molecule,breaking a bond. 2 1 3
Aldoses (Aldehyde Sugars) Ketoses (Ketone Sugars) Trioses: 3-carbon sugars (C3H6O3) Glyceraldehyde Dihydroxyacetone Pentoses: 5-carbon sugars (C5H10O5) Figure 5.3 Ribose Ribulose Hexoses: 6-carbon sugars (C6H12O6) Fructose Glucose Galactose
Ketose (Ketone Sugar) Aldose (Aldehyde Sugar) Trioses: 3-carbon sugars (C3H6O3) Figure 5.3a Glyceraldehyde Dihydroxyacetone
Ketose (Ketone Sugar) Aldose (Aldehyde Sugar) Pentoses: 5-carbon sugars (C5H10O5) Figure 5.3b Ribulose Ribose
Ketose (Ketone Sugar) Aldose (Aldehyde Sugar) Hexoses: 6-carbon sugars (C6H12O6) Figure 5.3c Galactose Fructose Glucose
6 6 1 2 5 5 3 4 1 4 1 4 2 2 Figure 5.4 5 3 3 6 (a) Linear and ring forms 6 5 4 1 2 3 (b) Abbreviated ring structure
1–4glycosidiclinkage 1 4 Figure 5.5 Glucose Glucose Maltose (a) Dehydration reaction in the synthesis of maltose 1–2glycosidiclinkage 1 2 Sucrose Glucose Fructose (b) Dehydration reaction in the synthesis of sucrose
Starch granules Chloroplast Amylopectin Figure 5.6 Amylose (a) Starch: a plant polysaccharide 1 m Glycogen granules Mitochondria Glycogen (b) Glycogen: an animal polysaccharide 0.5 m
Chloroplast Starch granules Figure 5.6a 1 m
Glycogen granules Mitochondria Figure 5.6b 0.5 m
(a) and glucose ring structures Figure 5.7 4 1 4 1 Glucose Glucose 4 1 1 4 (c) Cellulose: 1–4 linkage of glucose monomers (b) Starch: 1–4 linkage of glucose monomers
Figure 5.7a 1 1 4 4 Glucose Glucose (a) and glucose ring structures
1 4 Figure 5.7b (b) Starch: 1–4 linkage of glucose monomers 1 4 (c) Cellulose: 1–4 linkage of glucose monomers
Cellulosemicrofibrils in aplant cell wall Cell wall Microfibril 10 m Figure 5.8 0.5 m Cellulosemolecules Glucosemonomer
Cell wall Figure 5.8b 10 m
Figure 5.8c Cellulosemicrofibrils in a plant cell wall 0.5 m
The structureof the chitinmonomer Figure 5.9 Chitin forms the exoskeletonof arthropods. Chitin is used to make a strong and flexiblesurgical thread that decomposes after thewound or incision heals.
Figure 5.9a Chitin forms the exoskeletonof arthropods.
Figure 5.9b Chitin is used to make a strong and flexible surgical thread that decomposes after the wound or incision heals.
Fatty acid(in this case, palmitic acid) Glycerol Figure 5.10 (a) One of three dehydration reactions in the synthesis of a fat Ester linkage (b) Fat molecule (triacylglycerol)
Figure 5.10a Fatty acid(in this case, palmitic acid) Glycerol (a) One of three dehydration reactions in the synthesis of a fat
Ester linkage Figure 5.10b (b) Fat molecule (triacylglycerol)
(b) Unsaturated fat (a) Saturated fat Figure 5.11 Structuralformula of asaturated fatmolecule Structuralformula of anunsaturated fatmolecule Space-fillingmodel of stearicacid, a saturatedfatty acid Space-filling modelof oleic acid, anunsaturated fattyacid Cis double bondcauses bending.
(a) Saturated fat Figure 5.11a Structuralformula of asaturated fatmolecule Space-fillingmodel of stearicacid, a saturatedfatty acid
(b) Unsaturated fat Figure 5.11b Structuralformula of anunsaturated fatmolecule Space-filling modelof oleic acid, anunsaturated fattyacid Cis double bondcauses bending.
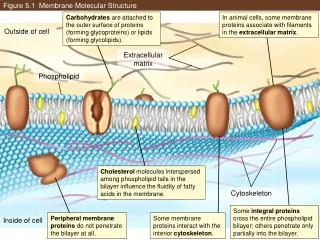
Choline Hydrophilic head Phosphate Figure 5.12 Glycerol Fatty acids Hydrophobic tails Hydrophilichead Hydrophobictails (a) Structural formula (b) Space-filling model (c) Phospholipid symbol
Choline Hydrophilic head Phosphate Glycerol Figure 5.12a Fatty acids Hydrophobic tails (a) Structural formula (b) Space-filling model
WATER Hydrophilichead Figure 5.13 Hydrophobictail WATER
Enzymatic proteins Defensive proteins Function: Protection against disease Function: Selective acceleration of chemical reactions Example: Digestive enzymes catalyze the hydrolysisof bonds in food molecules. Example: Antibodies inactivate and help destroyviruses and bacteria. Figure 5.15_a Antibodies Enzyme Virus Bacterium Storage proteins Transport proteins Function: Storage of amino acids Function: Transport of substances Examples: Hemoglobin, the iron-containing protein ofvertebrate blood, transports oxygen from the lungs toother parts of the body. Other proteins transportmolecules across cell membranes. Examples: Casein, the protein of milk, is the majorsource of amino acids for baby mammals. Plants havestorage proteins in their seeds. Ovalbumin is theprotein of egg white, used as an amino acid sourcefor the developing embryo. Transportprotein Amino acidsfor embryo Ovalbumin Cell membrane
Hormonal proteins Receptor proteins Function: Response of cell to chemical stimuli Function: Coordination of an organism’s activities Example: Receptors built into the membrane of anerve cell detect signaling molecules released byother nerve cells. Example: Insulin, a hormone secreted by thepancreas, causes other tissues to take up glucose,thus regulating blood sugar concentration Figure 5.15_b Receptorprotein Signalingmolecules Insulinsecreted Highblood sugar Normalblood sugar Structural proteins Contractile and motor proteins Function: Support Function: Movement Examples: Motor proteins are responsible for theundulations of cilia and flagella. Actin and myosinproteins are responsible for the contraction ofmuscles. Examples: Keratin is the protein of hair, horns,feathers, and other skin appendages. Insects andspiders use silk fibers to make their cocoons and webs,respectively. Collagen and elastin proteins provide afibrous framework in animal connective tissues. Actin Myosin Collagen Muscle tissue Connectivetissue 100 m 60 m
Enzymatic proteins Figure 5.15a Function: Selective acceleration of chemical reactions Example: Digestive enzymes catalyze the hydrolysisof bonds in food molecules. Enzyme
Storage proteins Function: Storage of amino acids Figure 5.15b Examples: Casein, the protein of milk, is the majorsource of amino acids for baby mammals. Plants havestorage proteins in their seeds. Ovalbumin is theprotein of egg white, used as an amino acid sourcefor the developing embryo. Amino acidsfor embryo Ovalbumin
Hormonal proteins Figure 5.15c Function: Coordination of an organism’s activities Example: Insulin, a hormone secreted by thepancreas, causes other tissues to take up glucose,thus regulating blood sugar concentration Insulinsecreted Highblood sugar Normalblood sugar
Contractile and motor proteins Function: Movement Examples: Motor proteins are responsible for theundulations of cilia and flagella. Actin and myosinproteins are responsible for the contraction ofmuscles. Figure 5.15d Actin Myosin Muscle tissue 100 m
Defensive proteins Figure 5.15e Function: Protection against disease Example: Antibodies inactivate and help destroyviruses and bacteria. Antibodies Virus Bacterium
Transport proteins Function: Transport of substances Figure 5.15f Examples: Hemoglobin, the iron-containing protein ofvertebrate blood, transports oxygen from the lungs toother parts of the body. Other proteins transportmolecules across cell membranes. Transportprotein Cell membrane
Receptor proteins Figure 5.15g Function: Response of cell to chemical stimuli Example: Receptors built into the membrane of anerve cell detect signaling molecules released byother nerve cells. Receptorprotein Signalingmolecules
Structural proteins Function: Support Examples: Keratin is the protein of hair, horns,feathers, and other skin appendages. Insects andspiders use silk fibers to make their cocoons and webs,respectively. Collagen and elastin proteins provide afibrous framework in animal connective tissues. Figure 5.15h Collagen Connectivetissue 60 m
Side chain (R group) Figure 5.UN01 carbon Aminogroup Carboxylgroup
Nonpolar side chains; hydrophobic Side chain(R group) Isoleucine (Ile or I) Glycine(Gly or G) Leucine(Leu or L) Alanine(Ala or A) Valine(Val or V) Figure 5.16 Methionine(Met or M) Phenylalanine(Phe or F) Tryptophan(Trp or W) Proline(Pro or P) Polar side chains; hydrophilic Threonine(Thr or T) Serine(Ser or S) Cysteine(Cys or C) Tyrosine(Tyr or Y) Asparagine(Asn or N) Glutamine(Gln or Q) Electrically charged side chains; hydrophilic Basic (positively charged) Acidic (negatively charged) Aspartic acid(Asp or D) Glutamic acid(Glu or E) Histidine(His or H) Lysine(Lys or K) Arginine(Arg or R)