Protein Structure and Function
E N D
Presentation Transcript
Proteins Topic 7.5
7.5 Proteins • 7.5.1 Explain the four levels of protein structure, indicating the significance of each level. • 7.5.2 Outline the difference between fibrous and globular proteins, with reference to two examples of each protein type. • 7.5.3 Explain the significance of polar and non-polar amino acids. • 7.5.4 State four functions of proteins, giving a named example of each.
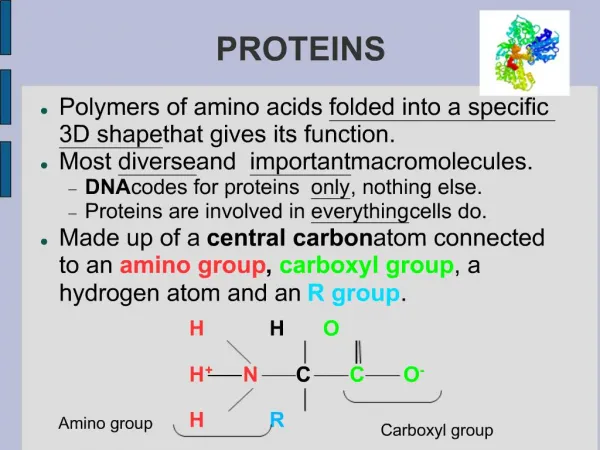
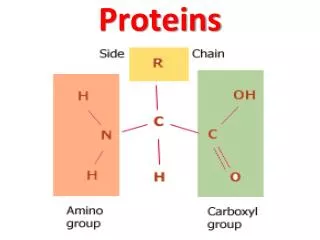
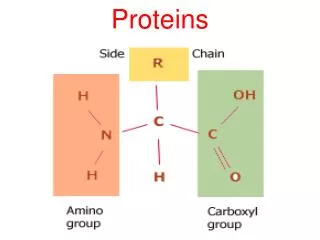
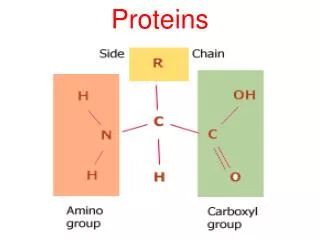
Four levels of protein structure • Primary organization • Chain of amino acids held together by polypeptide bonds • 20 amino acids may be arranged in any order and is determined by DNA • Primary structure determines the next three levels of protein organization • (Changing one amino acid here may completely alter the structure and function of a protein, as in sickle cell disease)
Secondary organization • Created by the formation of hydrogen bonds between the oxygen from the carboxyl group of one amino acid and the hydrogen from the amino group of another • Most common configurations • α-helix • β-pleated sheet
Tertiary organization • Polypeptide chain bends and folds over itself because of interactions among R-groups and the peptide backbone • Important in determining the specificity of the proteins known as enzymes • Interactions: • Disulfide bonds • Hydrogen bonds • Hydrophobic/hydrophillic • Ionic bonds
Quaternary organization • Involves multiple polypeptide chains which combine to form a single structure • Some include prosthetic or non-polypeptide groups called conjugatedproteins • Ex. Hemoglobin contains four polypeptide chains, each of which contains a non-polypeptide group called a heme (contains an iron atom that binds to oxygen)
Animations • http://www.johnkyrk.com/aminoacid.html
Fibrous and globular proteins • Fibrous proteins • Composed of many polypeptide chains in a long, narrow shape • Insoluble in water • Examples: • Collagen – structure of connective tissue in humans • Actin – component of human muscle • Globular proteins • More 3-D in their shape • Water soluble • Examples: • Hemoglobin – delivers oxygen to body tissues • Insulin – regulates blood glucose level in humans
Polar and non-polar amino acidsRefers to property of R group Polar Hydrophilic Found in regions exposed to water Membrane proteins toward interior and exterior of membrane Create hydrophilic channels in proteins through which polar substances can move Non-polar Hydrophobic Found in regions of proteins that are linked to the hydrophobic area of the cell membrane
Significance of polar and non-polar amino acids • Controls position of proteins in membranes, creating hydrophilic channels through membranes • Determinacy of specificity of an enzyme • Each enzyme has a region called the active site • Only specific substrates can combine with a particular active site • Combination is possible when ‘fitting’ occurs which involves general shape and polar properties of the substrate and the amino acids exposed at the active site