Chapter 5 Aromatic Compounds
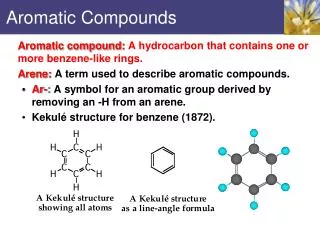
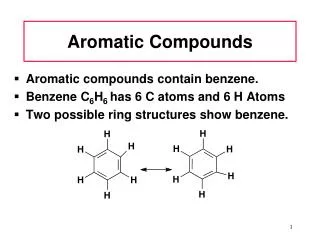
Chapter 5 Aromatic Compounds. 雌素酮. 5.1 Structure of Benzene: The KeKul’e Proposal. 1865. Born: Darmstadt, Germany, 7th September 1829. Died: Bonn, 13th July 1896. substitution. Benzene does not undergo electrophilic addition reactions. Why is benzene unreactive compared with other alkenes?

Chapter 5 Aromatic Compounds
E N D
Presentation Transcript
5.1 Structure of Benzene: The KeKul’e Proposal 1865 Born: Darmstadt, Germany, 7th September 1829.Died: Bonn, 13th July 1896. substitution Benzene does not undergo electrophilic addition reactions.
Why is benzene unreactive compared with other alkenes? • 2. Why does benzene give a substitution product rarher than addition product on reaction with Br2? • 3. All carbon-carbon bonds in benzene (139 pm) have the same length, intermediate between a typical single bond (154 pm) and a typical double bond (134 pm).
5.2 Structure of Benzene:The Resonance Proposal Bond length 0.139 nm Each carbon-carbon connection is an average of 1.5 bonds.
Monosubstituted Disubstituted
Benzenes with more than two substituents are named by numbering the position of each substituent on the ring so that the lowest possible numbers are used.
m-Bromochlorobenzene • Isobutylbenzene • P-Bromoaniline
5.4 Electrophilic Aromatic Substitution Reactions: Bromination Nucleophile Electrophile Halogenate: -F, -Cl, -Br, or –I Nitrate: -NO2 Sulfonate: -SO3H Alkylate: -R Acylate: -COR
Aromatic rings are less reactive toward electrophiles than alkenes are. The catalyst makes the Br2 molecule more electrophilic electrophile Nonaromatic carbocation intermediate Much less stable than benzene
Reaction of an electrophile with a benzene ring has relatively high activation energy and is rather slow.
5.5 Other Electrophilic Aromatic Substitution Reaction Chlorination 鎮靜安眠藥
Nitration nitronium ion
Limitation: • Only alkyl halides can be used; aryl halides such as chlorobenzene don’t react. • Do not succeed on aromatic rings that are already substituted by the groups –NO2, -CN, -SO3H, or –COR. Feiedel-Crafts acylation reaction
5.7 Substitutent Effects in Electrophilic Aromatic Substitution Substituents affect the reactivity of the aromatic ring
5.8 An Explanation of Substitutent Effects Activating and deactivating effects in aromatic rings: The common characteristic of all activating groups is that they donate electrons to the ring, stabilizing the carbocation intermediate.
Orienting effects in aromatic rings: ortho and para direction
5.11 Organic Synthesis Synthesize m-chloronitrobenzene starting from benzene