Lecture Notes for Chapter 15 Oxidative Phosphorylation
Essential Biochemistry Third Edition Charlotte W. Pratt | Kathleen Cornely. Lecture Notes for Chapter 15 Oxidative Phosphorylation. KEY CONCEPTS: Section 15-1.

Lecture Notes for Chapter 15 Oxidative Phosphorylation
E N D
Presentation Transcript
Essential Biochemistry Third Edition Charlotte W. Pratt | Kathleen Cornely Lecture Notes for Chapter 15 Oxidative Phosphorylation
KEY CONCEPTS: Section 15-1 • The standard reduction potential indicates a substance’s tendency to become reduced; the actual reduction potential depends on the concentrations of reactants. • Electrons are transferred from a substance with a lower reduction potential to a substance with a higher reduction potential. • The free energy change for an oxidation-reduction reaction depends on the change in reduction potential.
Recap on Oxidation-Reduction • One reactant is in its oxidized state while the other is in its reduced state. • Loss of electrons = oxidation • Gain of electrons = reduction
Reduction potential indicates a substance’s tendency to accept electrons.
The actual reduction potential depends on the actual concentrations of oxidized and reduced species. The Nernst Equation R = Gas Constant = 8.3145 J mol-1K-1T = temperature in Kelvin n = # of electrons F = Faraday’s constant = 96,485 J V-1K-1
The free energy change can be calculated from the change in reduction potential.
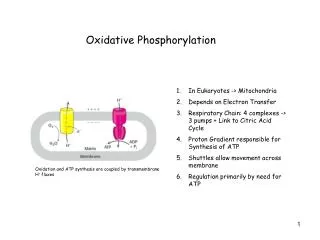
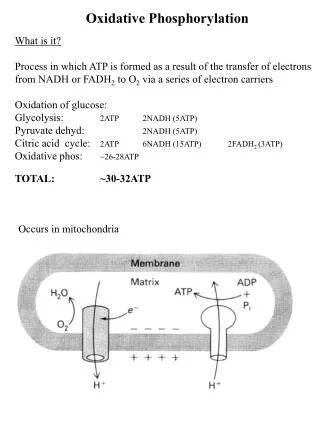
KEY CONCEPTS: Section 15-2 • The inner mitochondrial membrane encloses the matrix and includes specific transport proteins. • Complex I transfers electrons from NADH to ubiquinone. • The citric acid cycle, fatty acid oxidation, and other processes also generate mitochondrial ubiquinol. • The Q cycle mediated by Complex III reduces cytochrome c. • Complex IV uses electrons from cytochrome c to reduce O2 to H2O.
Experimental imaging helps us know what mitochondria look like. 3D Reconstruction by Electron Tomography Electron Micrograph of a Fibroblast Electron Micrograph Mitochondria in Green
The malate-aspartate shuttle transports reducing agents across the inner mitochondrial membrane.
A different transport system is used to move ATP from the matrix to the cytosol. A symport protein permitssimultaneous movement of Pi and H+. ATP translocase protein imports ADP and exports ATP.
Complex I binds ubiquinone. • Structure of a bacterial complex • FMN and Fe-S clusters are cofactors in space-filling representation. • Q binding site is highlighted. • Part of the protein has membrane-spanning a helices.
Complex I transfers electrons from NADH to Q. • Electrons transfer from NADH to FMN, then from FMN to Q. • As electrons are transferred from NADH to ubiquinone, Complex I transfers four protons from the matrix to the intermembrane space.
Iron-sulfur clusters undergo one-electron transfer reactions.
Other oxidation reactions contribute to the ubiquinol pool. • Succinate dehydrogenase produces QH2 during the citric acid cycle. • QH2 is also produced during fatty acid oxidation. • Electrons from cytosolic NADH can enter the mitochondrial ubiquinol pool through the actions of a cytosolic and a mitochondrial glycerol-3-phosphate dehydrogenase.
Complex III transfers electrons from ubiquinol to cytochrome c. • Cytochromes are proteins with heme prosthetic groups. • Unlike the heme groups in hemoglobin and myoglobin, heme in cytochrome c undergoes reversible one-electron transfers. • The central iron atom is either oxidized (Fe3+) or reduced (Fe2+).
Structure of Mammalian Complex III Cyt c1 Cyt b Arrangement of Prosthetic Groups Heme groups of Cyt b Fe-S clusters
The circuitous route of electrons from ubiquinol to cytochrome c is described by the Q cycle.
Results of the Q Cycle • Two electrons from QH2 reduce two molecules of cytochrome c. • Four protons are pumped into the intermembrane space. • Two from QH2 in the first round • Two from QH2 in the second round
Complex IV oxidizes cytochrome c and reduces O2. • Cytochrome c is a small membrane-soluble protein. • Cytochrome c transfers one electron at a time from Complex III to Complex IV. • Complex IV (cytochrome oxidase) catalyzes this reaction:
A Proposed Model for Cytochrome c Oxidase Activity • O2 is reduced to H2O via the Fe-S clusters in cytochrome c oxidase.
More on Complex IV Function • For every two electrons donated by cytochrome c, two protons are translocated to the intermembrane space • Two protons from the matrix are also consumed in the reaction: ½ O2 H2O
KEY CONCEPTS: Section 15-3 • The formation of a transmembrane proton gradient during electron transport provides the free energy to synthesize ATP. • Both concentration and charge contribute to the free energy of the proton gradient.
How much energy is available from electron transport? • This is enough energy to drive the endergonic phosphorylation of ADP to form ATP (ΔG°' = +30.5 kJ•mol–1)!
The imbalance of protons represents a source of free energy, also called a protonmotive force, that can drive the activity of an ATP synthase. [H+] = low [H+] = high
Computing the free energy change for the imbalance of protons. With respect to the electrical imbalance of protons: With respect to the chemical imbalance of protons: Z = Ion’s Charge Δy = Membrane Potential Combining these effects:
KEY CONCEPTS: Section 15-4 • Proton translocation drives the rotation of a portion of ATP synthase. • Rotation-induced conformational changes allow ATP synthase to bind ADP and Pi to phosphorylate ADP, and to release ATP. • Because ATP synthesis is indirectly linked to electron transport, the P:O ratio is not a whole number. • The supply of reduced cofactors determines the rate of oxidative phosphorylation.
The protein that taps the electrochemical proton gradient to phosphorylate ADP is known as ATP synthase (Complex V).
ATP Synthase Function Form ATP • Use theelectrochemicalgradient to drivephosphorylation.
ATP synthase rotates as it translocates protons. • H+ binds to a c subunit. • The c subunit moves away from the a subunit. • As a new c subunit reaches the a subunit a proton is released. • One rotation of the ring translocates 8 protons
The 3 ab pairs interact asymmetrically with the γ subunit. a = blue b = green g = purple
The binding change mechanism explains how ATP is made. • The ab subunits form three different conformations. • O = open • T = tight • L = loose • ADP and Pi bind to the open conformation. • ATP is formed in the tight conformation. • ATP is released in the open conformation.
Quantifying Oxidative Phosphorylation • The P:O ratio describes the stoichiometry of oxidative phosphorylation. • P:O ratio = # phosphorylations of ADP per # of oxygen atoms reduced • P:O ratios will not be integral because chemical energy becomes a protonmotive force, then mechanical movement of ATP synthase and back to chemical energy • The rate of oxidative phosphorylation depends on the rate of fuel catabolism. • Oxidative phosphorylation is regulated by the availability of reduced cofactors (NADH and QH2) produced by other metabolic processes.