Proteins
Proteins. Proteins are long polymers made up of 20 different amino acid monomers They are quite large, with molar masses of around 5,000 g/mol to around 100,000 g/mol They have complex structures with unique 3-D shapes that determine their functions

Proteins
E N D
Presentation Transcript
Proteins • Proteins are long polymers made up of 20 different amino acid monomers • They are quite large, with molar masses of around 5,000 g/mol to around 100,000 g/mol • They have complex structures with unique 3-D shapes that determine their functions • They are the most abundant organic compounds in the body, and also the most diverse in function • Proteins are involved in structure, transport, storage, metabolism, cell signaling and many other processes
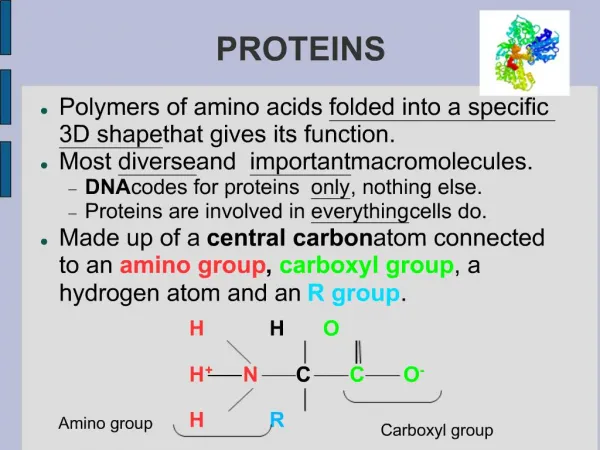
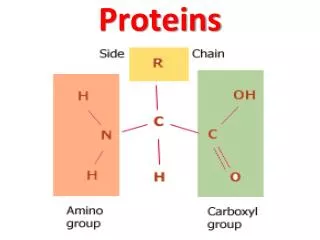
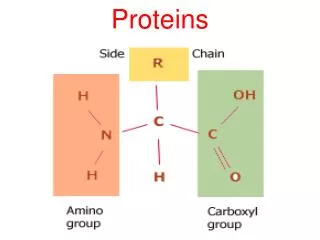
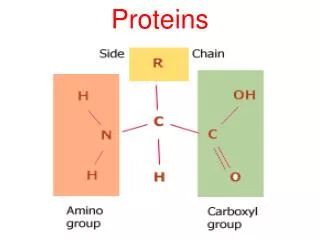
Amino Acids • Amino acids, as the name implies, have both an amine group and a carboxylic acid group • The 20 amino acids that make up our proteins have the amine group, the acid group, a hydrogen, and a variable group attached to a central carbon (called the carbon) • The variable groups (called side chains) are what determine the individual characteristics of the amino acids General structure of an amino acid:
Acidic and Basic Amino Acids • Amino acids can be classified by the nature of the side-chain as acidic, basic, polar neutral or nonpolar
Abbreviations for Amino Acids • Each amino acid has standard 3-letter and 1-letter abbreviations (shown in the table below)
D- and L-Amino Acids • All amino acids besides glycine are chiral • Each amino acid has two possible enantiomers - these are classified as D or L as with sugars • Amino acids in nature are almost exclusively L-amino acids • When a Fischer projection is written with the acid at the top, and the R group at the bottom: - if the amine group is on the right, it’s a D-amino acid - if the amine group is on the left, it’s an L-amino acid
Isoelectric Points for Amino Acids • Because the amine group is basic, and the carboxylic acid group is acidic, amino acids often exist as zwitterions • A zwitterion is a dipolar ion with a net charge of zero • Because zwitterions act like salts, they have high melting points • The isoelectric point (pI) is the pH at which a zwitterion forms - below pI the amino acid has a net positive charge - above pI the amino acid has a net negative charge • Acidic amino acids have low pI values and basic amino acids have high pI values (due to side-chain ionization)
Electrophoresis of Amino Acids • Electrophoresis is a technique used to separate charged molecules with an electric field • The samples are loaded onto a support medium (usually an agarose or polyacrylamide gel) and separated by mobility - mobility is affected by size, shape, charge and solubility • A buffered solution is used to conduct the charge and allow the charged molecules to move - negatively charged amino acids move towards the anode (-) - positively charged amino acids move towards the cathode (+)
Peptides • Peptides are two or more amino acids linked together by amide bonds (called peptide bonds) • A peptide bond is formed when the acid group of one amino acid reacts with the amine group of another amino acid • When writing the structure of a peptide: - the amino acid with the free (unreacted) amine group is written on the left and is called the N terminal amino acid - the amino acid with the free (unreacted) acid group is written on the right and is called the C terminal amino acid • Peptides are usually named using the 3- or 1-letter abbreviations for the amino acids, going from N terminal to C terminal
Synthesis of Peptides and Proteins • In cells, peptides and proteins are synthesized using RNA catalysts (to be discussed in Chapter 22) • In the laboratory a variety of techniques are used - most commonly the peptides are synthesized on resin beads using an automated peptide synthesizer - smaller peptides, like dipeptides, are generally synthesized by hand in solution (not on resin) - protecting groups must be used in order to prevent unwanted amino acid couplings
Structure of Peptide Bonds • Peptides are particularly stable and are also fairly rigid • This is due to the structure of the peptide amide bonds • Through resonance, the lone pair electrons on nitrogen and the pi electrons of the carbonyl are delocalized - this gives some double bond character to the C-N bond, preventing free rotation around that bond - this also makes the nitrogen less basic, since the lone pair is not very available for bonding, increasing peptide stability
Primary Structure of Peptides and Proteins • A polypeptide containing 50 or more amino acids is usually called a protein • The primary structure of a protein is the sequence of amino acids in the peptide chain • The higher levels of structure, as well as the function, are derived from the primary structure - even a single amino acid change can have drastic effects • For example, the nonapeptides oxytocin and vasopressin only differ in the amino acids at positions 3 and 8
Insulin • Insulin was the first protein whose primary structure was determined • Human, pig and cow insulin differ only at four amino acids • Bovine insulin (from cow pancreas) was used for diabetics, but now it’s made by genetically engineered E. coli
Secondary Structure of Proteins (the Alpha Helix) • The secondary structure of a protein indicates the conformation of the peptide chain in a given region • There are three main types of secondary structure: the alpha helix, the beta-pleated sheet and the triple helix - all three are governed by hydrogen bonding • The alpha helix is coiled due to H-bonding between backbone N-H on one loop to backbone C=O group on next loop • The side chains are all on the outside of the helix, so larger side chain groups favor helix
Secondary Structure of Proteins (the Beta-Pleated Sheet) • Beta-pleated sheets consist of peptide chains side-by-side, held together by backbone H-bonding • All the side chains point out above and below the sheet - smaller side chains favor -pleated sheets (larger ones would be too crowded)
Secondary Structure of Proteins (the Triple Helix) • A triple helix consists of three peptide strands in a braid, held together by H-bonding, both backbone H-bonding and H-bonding between hydroxyl groups on adjacent peptide strands - they contain large amounts glycine, proline, hydroxyproline and hydroxylysine that contain –OH groups for H-bonding • Triple helices are very strong, and are found in collagen, connective tissue, skin, tendons, and cartilage - several triple helices can form a larger braid for increased strength