Auger Electron Spectroscopy
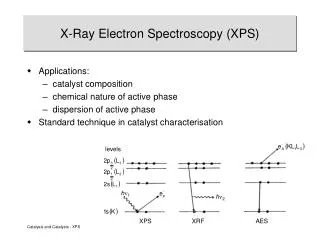
Auger Electron Spectroscopy. (AES). Principle of AES. A fine focused electron beam bombards the sample and ejects an electron of the inner shell of the atom. This vacancy must be refilled by an electron from a higher energy level.

Auger Electron Spectroscopy
E N D
Presentation Transcript
Principle of AES • A fine focused electron beam bombards the sample and ejects an electron of the inner shell of the atom. • This vacancy must be refilled by an electron from a higher energy level. • When the higher energy electron fills the hole, the release of energy is transferred to an electron in an outer orbit electron. • That electron has sufficient energy to overcome the binding energy and the work function to be ejected with a characteristic kinetic energy. • The ejected electron is referred to as an Auger electron after Pierre Auger who first discovered it in 1925.
Principle of AES • The Auger electron has an energy given by: EAuger = EK – EL1 – EL2,3 - where EK, EL1 and EL2,3 are the binding energies of the K1, L1 and L2,3 electron orbits of the atom. is the work function.
Cylindrical Mirror Analyzer (CMA) • Secondary electrons passing through a slit in the inner cylinder are deflected by a negative potential applied to the outer cylinder and pass through the exit slit onto an electron multiplier where they are detected. • For any given potential applied to the cylinder, only electrons with specific energy will pass through the exit slit. • A spectrum is built up by sweeping the outer cylinder potential.
References • J. M. Walls, Editor, Methods of Surface Analysis: Techniques and Applications, Cambridge University Press, Cambridge, 1989. • J. P. Sibilia, Editor, A Guide to Materials Characterization and Chemical Analysis, VCH Publishers, Inc., New York, 1988. • P. E. J. Flewitt and R. K. Wild, Physical Methods for Materials Characterization, Institute of Physics Publishing, Bristol, 1994. • D. Briggs and M. P. Seah, Practical Surface Analysis by Auger and X-ray Photoelectron Spectroscopy, John Wiley and Sons, New York, 1983.