Haemoglobin Dissociation Curve for Oxygen Transport
Explore how haemoglobin carries oxygen, significance of dissociation curves, ways carbon dioxide is transported in blood, and effects of CO2 concentration on oxygen affinity.

Haemoglobin Dissociation Curve for Oxygen Transport
E N D
Presentation Transcript
Settler Using your models from last lesson take it in turns to explain to each the shape of the dissociation curve. Include keywords Oxyhaemoglobin Haem Affinity Oxygen tension Diffusion gradient Conformational change
Module 3Exchange and transport 8.4 Carriage of carbon dioxide
Starter • Write down both the word and symbol equation for respiration
Success Criteria Learning Objectives To understand how haemoglobin carries oxygen Describe and explain the significance of the dissociation curves of adult oxyhaemoglobin at different carbon dioxide levels List the ways carbon dioxide can be transported by the blood (Grade E - D) Describe the role of haemoglobin in carrying carbon dioxide (Grade C –B) Describe and explain how haemoglobin can supply more oxygen to actively respiring tissues than to those that have a lower level of respiration (Grade B – A)
How is carbon dioxide transported? • Carbon dioxide is released from respiring tissues and transported to the lungs in 3 different ways • 85% transported in the form of Hydrogencarbonate - HCO3− which will eventually diffuse back into the plasma • 10% combined directly with haemoglobin to form carbaminohaemoglobin • 5% dissolved directly into plasma • List the ways carbon dioxide can be transported by the blood (Grade E - D)
Explanation of Bohr Effect Carbon dioxide diffuses into the blood combining with water forming carbonic acid, catalysed by enzyme carbonic anhydrase. Carbonic acid dissociates to release H+ and HCO3− • CO2 + H2O H2CO3 H+ + HCO3− carbonic acid hydrogen ions +hydrogen carbonate ions HCO3− diffuses out of RBC s and into plasma. The charge inside the RBC is maintained by the movement of chloride ions from the plasma into the RBC (chloride shift) H+ are taken up by haemoglobin to produce HHb, to maintain pH • Hb + H+ Haemoglobinic acid • H+ cause acidity that promotes oxygen release
Describe the role of haemoglobin in carrying carbon dioxide (Grade C –B) Task Using information from the lesson complete the card sort demonstrating the Bohr effect H2CO3 Carbonic acid HCO3− hydrogencarbonate ions HHb haemoglobinic acid (haemoglobin acts as a buffer, formed when oxyhaemoglobin combines with hydrogen ions)
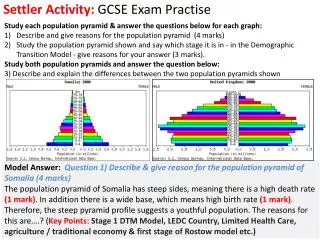
Effects of Carbon Dioxide Concentration • How do you think CO2 will affect the affinity of haemoglobin for oxygen? • At a high CO2 concentration: Haemoglobin has a reduced affinity for oxygen • At a low CO2 concentration: Haemoglobin has a higher affinity for oxygen Bohr Effect CO2+ H2O H+ + HCO-3
Describe and explain how haemoglobin can supply more oxygen to actively respiring tissues than to those that have a lower level of respiration(Grade B – A) Effects of Carbon Dioxide Concentration • Hb gives up its oxygen more readily at higher partial pressures of carbon dioxide. 1. Cells respire producing CO2 this raises p CO2 2. This increases the rate of oxygen unloading (dissociation curve shifts down)The oxygen is lower for a given pO2 meaning more oxygen is released • The Bohr Effect – graph shifts to the right • TASK – Describe and explain the significance of the dissociation curves of adult oxyhaemoglobin at different carbon dioxide levels
Answers Questions: • How does this explain behaviour of haemoglobin in the lungs? • How does this explain behaviour of haemoglobin in the muscles? • Why does haemoglobin have a reduced affinity for oxygen at higher CO2 concentrations? • In the lungs the level of carbon dioxide is low because it diffuses across the surface and is breathed out. So, haemoglobin has a higher affinity for oxygen (there is also a higher concentration of oxygen in the lungs). This means oxygen is readily loaded by haemoglobin. • In the muscles the level of carbon dioxide is high because it is produced by respiration. So, haemoglobin has a lower affinity for oxygen (there is also a lower concentration of oxygen in the muscles). This means oxygen is readily unloaded by haemoglobin. • Dissolved CO2 is acidic and the low pH causes haemoglobin to change shape, making it readily lose its oxygen when there is a high concentration of CO2. CO2+ H2O H+ + HCO-3
Task • Answer questions 1- 5 – Homework complete • Revise all sections – animal transport – 1-12 in the book for test next lesson
100 90 80 70 60 50 40 30 20 10 0 0 2 4 6 8 10 12 14 What is myoglobin? Myoglobin is a molecule with a similar structure to haemoglobin, but with only one haem group. Myoglobin has a very high affinity for oxygen, even at very low partial pressures. myoglobin haemoglobin oxyhaemoglobin (% saturation) This means oxymyoglobin will only dissociate when oxygen levels are low. It is found in muscle cells, where it acts as an oxygen reserve. oxygenpartial pressure (kPa)