Proteins
Proteins. Proteins have many structures , resulting in a wide range of functions Proteins do most of the work in cells and act as enzymes Proteins are made of monomers called amino acids. Table 5.1. An overview of protein functions. Substrate binds to enzyme.

Proteins
E N D
Presentation Transcript
Proteins • Proteins have many structures, resulting in a wide range of functions • Proteins do most of the work in cells and act as enzymes • Proteins are made of monomers called amino acids
Table 5.1 • An overview of protein functions
Substrate binds to enzyme. 1 Active site is available for a molecule of substrate, the reactant on which the enzyme acts. 2 2 Substrate (sucrose) Glucose Enzyme (sucrase) OH H2O Fructose H O 4 Products are released. 3 Substrate is converted to products. Figure 5.16 • Enzymes • Are a type of protein that acts as a catalyst, speeding up chemical reactions
Polypeptides • Polypeptides • Are polymers (chains) of amino acids • A protein • Consists of one or more polypeptides
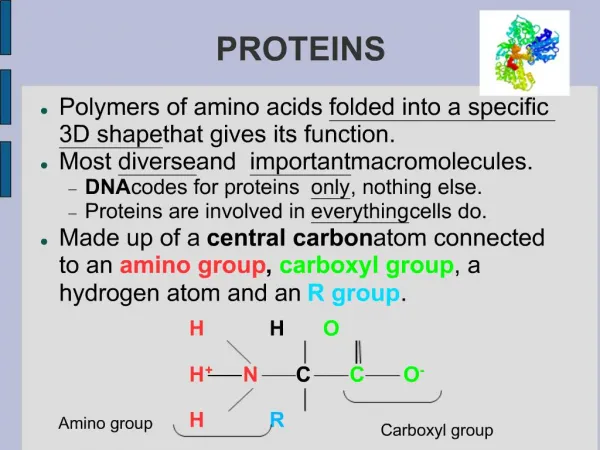
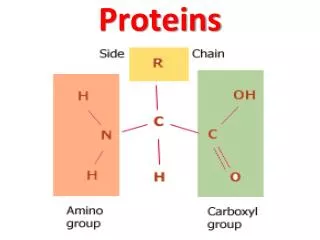
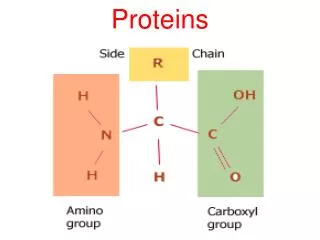
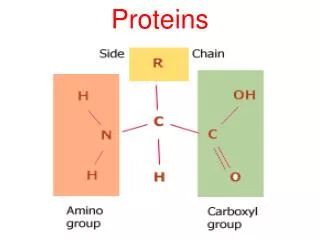
Amino acids • Are organic molecules possessing both carboxyl and amino groups • Differ in their properties due to differing side chains, called R groups
CH3 CH3 CH3 CH CH2 CH3 CH3 H CH3 H3C CH3 CH2 CH O O O O O H3N+ H3N+ H3N+ H3N+ C H3N+ C C C C C C C C C O– O– O– O– O– H H H H H Valine (Val) Leucine (Leu) Isoleucine (Ile) Glycine (Gly) Alanine (Ala) Nonpolar CH3 CH2 S H2C CH2 O NH CH2 H2N C C CH2 CH2 O– CH2 O O O H H3N+ H3N+ C C C C H3N+ C C O– O– O– H H H Phenylalanine (Phe) Proline (Pro) Methionine (Met) Tryptophan (Trp) Figure 5.17 Twenty Amino Acids • 20 different amino acids make up proteins
OH NH2 O C NH2 O C OH SH CH2 CH3 OH Polar CH2 CH CH2 CH2 CH2 CH2 O O O O O O H3N+ H3N+ H3N+ H3N+ H3N+ H3N+ C C C C C C C C C C C C O– O– O– O– O– O– H H H H H H Glutamine (Gln) Tyrosine (Tyr) Asparagine (Asn) Cysteine (Cys) Serine (Ser) Threonine (Thr) Basic Acidic NH3+ NH2 NH+ O– O –O O CH2 C NH2+ C C NH Electrically charged CH2 CH2 CH2 CH2 CH2 O O H3N+ H3N+ CH2 CH2 C CH2 C C C O O– H3N+ O– CH2 C CH2 C H O H H3N+ O– C C CH2 H O O– H3N+ C C H O– H Lysine (Lys) Histidine (His) Arginine (Arg) Glutamic acid (Glu) Aspartic acid (Asp)
Amino Acid Polymers • Amino acids • Are linked by peptide bonds
Protein Conformation and Function • A protein’s specific conformation (shape) determines how it functions
Amino acid subunits +H3NAmino end Pro Thr Gly Gly Thr Gly Glu Seu Lys Cys Pro Leu Met Val Lys Val Leu Asp Ala Arg Val Gly Ser Pro Ala Glu Lle Asp Thr Lys Ser Tyr Trp Lys Ala Leu Gly lle Ser Pro Phe His Glu His Ala Glu Val Thr Phe Val Ala Asn lle Thr Asp Ala Tyr Arg Ser Ala Arg Pro Gly Leu Leu Ser Pro Tyr Ser Tyr Ser Thr Thr Ala o Val c Val Glu – Lys o Thr Pro Asn Carboxyl end Figure 5.20 Four Levels of Protein Structure • Primary structure • Is the unique sequence of amino acids in a polypeptide
H H H H H H O O O O O O O H H H H H H R R R R R R R C C C C C C C C C C C C C N N N N N N N N N N N N N C C C C C C C C C C C C C C R R R R R R H H H H H H H O O O O O O O H H H H H H H pleated sheet H O H H Amino acidsubunits C C N N N C C C R H O H H H H H H N N N N N N helix C C O C H H H C C C R R R R R H H C C C C C C O O O O H C R O C C O H C O N N H C C H R H R Figure 5.20 • Secondary structure • Is the folding or coiling of the polypeptide into a repeating configuration • Includes the helix and the pleated sheet
Hydrophobic interactions and van der Waalsinteractions CH CH2 CH2 H3C CH3 OH Polypeptidebackbone H3C CH3 Hyrdogenbond CH O HO C CH2 CH2 S S CH2 Disulfide bridge O -O C CH2 CH2 NH3+ Ionic bond • Tertiary structure • Is the overall three-dimensional shape of a polypeptide • Results from interactions between amino acids and R groups
Polypeptidechain Collagen Chains Iron Heme Chains Hemoglobin • Quaternary structure • Is the overall protein structure that results from the aggregation of two or more polypeptide subunits
+H3N Amino end Amino acid subunits helix Review of Protein Structure
Sickle-Cell Disease: A Simple Change in Primary Structure • Sickle-cell disease • Results from a single amino acid substitution in the protein hemoglobin
Normal hemoglobin Sickle-cell hemoglobin Primary structure Primary structure . . . . . . Exposed hydrophobic region Val His Leu Thr Pro Glul Glu Val His Leu Pro Glu Thr Val 5 6 7 3 4 5 6 7 1 2 1 2 3 4 Secondaryand tertiarystructures Secondaryand tertiarystructures subunit subunit Quaternary structure Hemoglobin A Quaternary structure Hemoglobin S Molecules interact with one another tocrystallize into a fiber, capacity to carry oxygen is greatly reduced. Function Molecules donot associatewith oneanother, eachcarries oxygen. Function 10 m 10 m Normal cells arefull of individualhemoglobinmolecules, eachcarrying oxygen Red bloodcell shape Red bloodcell shape Figure 5.21 Fibers of abnormalhemoglobin deform cell into sickle shape.
What Determines Protein Conformation? • Protein conformation Depends on the physical and chemical conditions of the protein’s environment • Temperature, pH, etc. affect protein structure
Denaturation Normal protein Denatured protein Renaturation Figure 5.22 Denaturation is when a protein unravels and loses its native conformation(shape)
The Protein-Folding Problem • Most proteins • Probably go through several intermediate states on their way to a stable conformation • Denaturated proteins no longer work in their unfolded condition • Proteins may be denaturated by extreme changes in pH or temperature
Correctlyfoldedprotein Polypeptide Cap Hollowcylinder The cap attaches, causing the cylinder to change shape insuch a way that it creates a hydrophilic environment for the folding of the polypeptide. The cap comesoff, and the properlyfolded protein is released. Steps of ChaperoninAction: An unfolded poly- peptide enters the cylinder from one end. Chaperonin(fully assembled) 2 3 1 Figure 5.23 • Chaperonins • Are protein molecules that assist in the proper folding of other proteins
X-raydiffraction pattern Photographic film Diffracted X-rays X-ray beam X-raysource Crystal Nucleic acid Protein (b) 3D computer model (a) X-ray diffraction pattern • X-ray crystallography • Is used to determine a protein’s three-dimensional structure Figure 5.24
Nucleic Acids • Nucleic acids store and transmit hereditary information • Genes • Are the units of inheritance • Program the amino acid sequence of polypeptides • Are made of nucleotide sequences on DNA
The Roles of Nucleic Acids • There are two types of nucleic acids • Deoxyribonucleic acid (DNA) • Ribonucleic acid (RNA)
Deoxyribonucleic Acid • DNA • Stores information for the synthesis of specific proteins • Found in the nucleus of cells
DNA 1 Synthesis of mRNA in the nucleus mRNA NUCLEUS CYTOPLASM mRNA 2 Movement of mRNA into cytoplasm via nuclear pore Ribosome 3 Synthesis of protein Aminoacids Polypeptide Figure 5.25 DNA Functions • Directs RNA synthesis (transcription) • Directs protein synthesis through RNA (translation)
5’ end 5’C O 3’C O O 5’C O 3’C 3’ end OH Figure 5.26 The Structure of Nucleic Acids • Nucleic acids • Exist as polymers called polynucleotides (a) Polynucleotide, or nucleic acid
Nucleoside Nitrogenous base O 5’C O O CH2 P O O Phosphate group 3’C Pentose sugar Figure 5.26 (b) Nucleotide • Each polynucleotide • Consists of monomers called nucleotides • Sugar + phosphate + nitrogen base
Nitrogenous bases Pyrimidines NH2 O O C C CH3 C N CH HN C CH HN CH CH C CH C C CH CH N N O N O O H H H Cytosine C Uracil (in RNA) U Thymine (in DNA) T Uracil (in RNA) U Purines O NH2 C C N N C C NH N HC HC C CH C N N NH2 N N H H Adenine A Guanine G Pentose sugars 5” 5” OH OH HOCH2 HOCH2 O O H H H H 1’ 1’ 4’ 4’ H H H H 3’ 2’ 3’ 2’ H OH OH OH Deoxyribose (in DNA) Ribose (in RNA) Ribose (in RNA) Nucleotide Monomers • Nucleotide monomers • Are made up of nucleosides (sugar + base) and phosphate groups Figure 5.26 (c) Nucleoside components
Nucleotide Polymers • Nucleotide polymers • Are made up of nucleotides linked by the–OH group on the 3´ carbon of one nucleotide and the phosphate on the 5´ carbon on the next
Gene • The sequence of bases along a nucleotide polymer • Is unique for each gene
The DNA Double Helix • Cellular DNA molecules • Have two polynucleotides that spiral around an imaginary axis • Form a double helix
3’ end 5’ end Sugar-phosphatebackbone Base pair (joined byhydrogen bonding) Old strands Nucleotideabout to be added to a new strand 3’ end A 5’ end Newstrands 3’ end 3’ end 5’ end Figure 5.27 • The DNA double helix • Consists of two antiparallel nucleotide strands
A,T,C,G • The nitrogenous bases in DNA • Form hydrogen bonds in a complementary fashion (A with T only, and C with G only)
DNA and Proteins as Tape Measures of Evolution • Molecular comparisons • Help biologists sort out the evolutionary connections among species
The Theme of Emergent Properties in the Chemistry of Life: A Review • Higher levels of organization • Result in the emergence of new properties • Organization • Is the key to the chemistry of life