Understanding Atoms, Minerals, and Rocks: Earth's Building Blocks
240 likes | 345 Vues
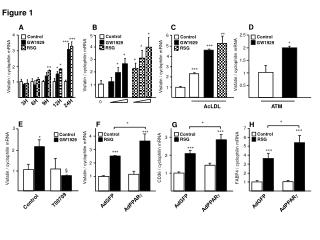
Explore geology, rocks, minerals, and Earth processes. Learn about atoms, elements, compounds, and bonding types in forming minerals. Discover the composition and classification of mafic and felsic silicates.

Understanding Atoms, Minerals, and Rocks: Earth's Building Blocks
E N D
Presentation Transcript
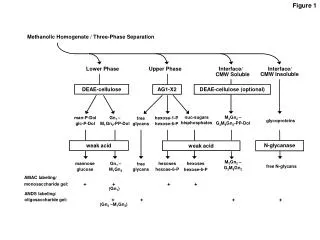
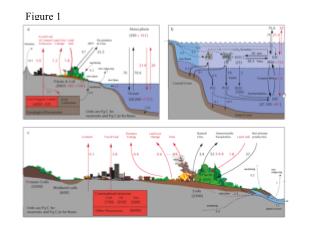
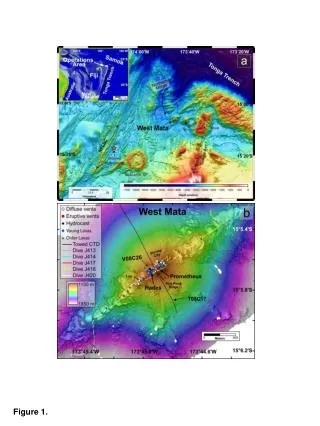
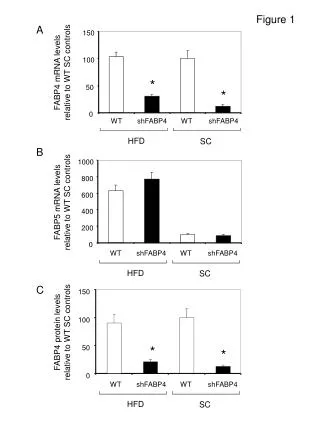
Atoms and Minerals Building blocks for Rocks and the Earth Figure 1, p. 76
Geology- Study of the Earth Rocks - What are they made of? How do they form and change? What do the tell us about the Earth and Earth processes? What is a Mineral? Matter - What is it?
Atoms Nucleus Protons (+) Neutrons Electrons (-) Fig. 3-2, p. 61
No of Protons Table 3-1, p. 62
Elements combine to form Compounds BONDING Ionic: Metals lose electron (Na+,K+, Ca++) nonmetals gain electron (Cl-, F-) NaCl, CaF2 Covalent: shared electrons, each atom donates electron to bond (e.g., diamond)
Ionic Bonding Fig. 3-4, p. 63
Covalent Bonding Diamond Graphite Covalent within sheets van der Waals forces between sheets Fig. 3-5, p. 64
OTHER TYPES OF BONDING: Metallic: positive ions (lost electrons), shared common electron “cloud” electrons can "flow" (electricity) e.g., Copper Van der Waals Forces: weak, attraction between charged molecules (e.g., water, H2O - H2O
MINERAL Naturally Occurring Inorganic Shells OK (calcite, aragonite; CaCO3) NOT coal, oil, or amber Crystalline (Not petroleum, obsidian) Restricted chemical composition
What are the two most abundant elements in the Earth’s crust?
The Building Block of Silicate Rocks: The Silica Tetrahedron Fig. 3-10, p. 69
Mafic Silicates Felsic Limestone Evaporites Table 3-4, p. 79
Mafic Felsic Fig. 3-12, p. 71
Things to Know: Atom Mineral 2 most abundant elements in Earth’s crust “Building block” of silicate minerals =silica tetrahedron Mafic vs Felsic silicates Examples of mafic and felsic minerals Others: Limestone (Calcium Carbonate), Evaporites (salt, gypsum)