Advanced Recombineering Techniques in Molecular Biology
220 likes | 272 Vues
Explore the efficient engineering of PCR products in E. coli using homologous recombination technology explained in detail, with references and practical applications. Learn the mechanisms and tools like RecA, Red system, and GalK for targeted DNA manipulation. Discover the comparison to traditional genetic engineering methods and practical applications in plasmid DNA amplification and gene editing.

Advanced Recombineering Techniques in Molecular Biology
E N D
Presentation Transcript
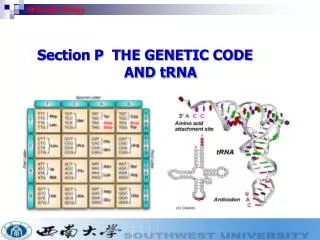
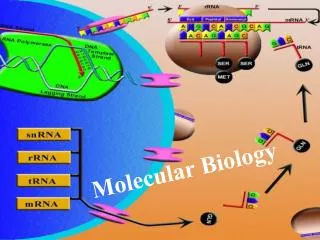
Methods in Molecular Biology Recombineering Ólafur S. Andrésson 13th October 2005
Recombineering: Engineered homologous recombination of PCR products or oligos in E. coli. Analogous to homologous recombination technology in S. cerevisiae and S. pombe. References (read!): Court, DL, Sawitzke, JA,Thomason, LC. 2002. Genetic engineering using homologous recombination. Ann. Rev. Genet. 36:361-388. And chapter from Current Protocols in Molecular Biology. See also: http://recombineering.ncifcrf.gov/
E. coli homologous recombination is dependent on recA. RecBCD generates 3´single strand overhangs. RecA binds ssDNA and mediated strand invasion. RecF-pathway similar but more complex. Acts primarily at replication forks.
Phage recombination systems not dependent on recA: λ phage Red functions: Exo, Beta, Gam. Rac prophage functions: RecE and RecT.
According to this model Red-mediated homologous recombination should be more efficient with homologous ends than with end + linear DNA. Does not appear to be much of a hindrance – we have used Red system to recombine into plasmid.
λ phage Red functions: Exo, Beta, Gam. Gaminhibits two nucleases, RecBCD and SbcCD both involved in double-srand break dependent recombination. RecBCD and SbcCD destroy linear dsDNA. Coordinate expression of Exo, Beta and Gam.
λ Exo: 5´to 3´dsDNA-dependent exonuclease. λ Beta: ssDNA-binding and anneals complementary strands.
In vivo cloning by Gap-Repair in E. coli. Originally done in recBC sbC strains – enhanced by Red or RecET (Rac) systems.
Cloning PKS orfs – removal of introns 6-MSAS gene P. patulum chromosome stýrill Amplification of fragments: Recombination in yeast Plasmid DNA Amplification performed with Taq or Dynazyme with Pfu-Ultra Has been used to assemble 9-10 kb PKS genes and remove 5-6 introns at the same time.
Comparison of standard genetic Engineering and recombineering.
Model for RecA- independent recombination of dsDNA at replication fork.
Model for RecA-independent recombination of dsDNA cassette: Two replication forks.
GalK allows 1) selection and 2) counter- selection. 1) ∆galK host 2) DOG 2-deoxygalactose selects against GalK+ cells
Selective marker can be targeted to any sequence in E. coli by recombineering.
LAB 21st October: 1) Insertion of KanR cassette at end of E. coli lig gene (encoding DNA ligase). Producing deletion of carboxy-terminal domain of ligsase enzyme. 2) Insertion of ZeoR cassette at end of 9 kb lichen PKS gene in 15 kb plasmid. Adding a 6Xhis tail on PKS enzyme to be able to detect protein in transformed host.